acid in soap, check these out | Is there acid in soap?
Acids commonly added to soap include lemon juice (citric acid), vinegar (acetic acid), and yogurt (lactic acid). Adding these acids complicate the soap making process, because they react faster and more easily with NaOH or KOH than fatty acids can react.
Is there acid in soap?
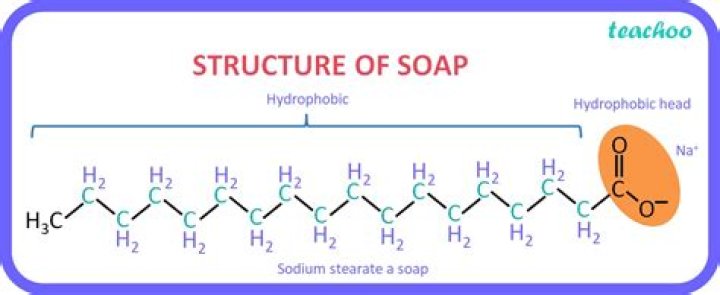
Soap is a combination of a weak acid (fatty acids) and a strong base (lye), which results in what is known as “alkalai salt,” or a salt that is basic on the pH scale.
What acid is used in soap and detergent?
To protect skin and other materials from the abrasive effects of lye, it is necessary to add fat in the forms of oils and butters to soap recipes. In this process, called saponification, triglycerides react with lye to produce glycerol and a fatty acid (soap).
Why is fatty acid in soap?
Saponification is the process in which triglycerides are combined with a strong base to form fatty acid metal salts during the soap-making process. The distribution of unsaturated and saturated fatty acid determines the hardness, aroma, cleansing, lather, and moisturizing abilities of soaps.
How much acid does soap have?
The average percentage of ricinoleic acid in the favorite soap recipes of soapmakers polled rounds in at 5%. Most recipes clocked in at 4% to 7% ricinoleic acid. My favorite formulas tend to fall in the high end of that range and above, between 6% and 10% ricinoleic acid. Yup, I love me some castor oil in soap.
What happens if you add acid to soap?
The added stronger acid will still have a bullying nature when mixed with finished soap. It will bump the weaker fatty acid out of its place on the soap molecule and take its place. The altered molecule will then be the salt of the stronger acid.
What pH is soap?
Results: Majority of the soaps have a pH within the range of 9-10. Majority of the shampoos have a pH within the range of 6-7. Conclusions: The soaps and shampoos commonly used by the population at large have a pH outside the range of normal skin and hair pH values.
What are the 3 main ingredients in soap?
If you only rinse them with water, they still feel greasy. However, if you add soap to the water, the grease washes away, and the result is clean dishes. There are 3 key ingredients in soap: oil or fat, lye and water.
What are the active ingredients in soap?
There are two types of lye used by soapmakers – sodium hydroxide and potassium hydroxide. Sodium hydroxide is used to make solid soap; potassium hydroxide is used to make liquid soaps.
Can soap be made without lye?
The main way that you can make soap without handling lye is by using melt-and-pour soap. It’s already been through saponification (oils reacting with lye) and is safe to use and handle straight out of the package. All you do with it is melt it, add your scent, color, and other additives, then pour it into molds.
Why is lauric acid in soap?
Lauric acid in soap is a cleaning powerhouse—when used at very high ratios, it can over cleanse the skin, leaving it dry. Soap making oils high in lauric acid add hardness and a good fluffy lather to soap bars. The unique fatty acid profile of the oils you use in your batch of soap affects the finished soap.
What is lauric acid in soap?
Lauric acid is a saturated fat and a medium-chain fatty acid, made up of a twelve atom carbon chain. What does this mean to you? Oils high in lauric acid will make soap that gets hard and offers mounds of fluffy bubbles for your bathing pleasure. It also creates a super cleansing bar.
What is coconut acid in soap?
What Does Coconut Fatty Acid Do in Our Products? Coconut fatty acid is surfactant or cleansing agent. [4,5] It is often found in laundry and dishwashing products, soap, face cleansers, shampoo, deodorant, body wash, and other products.
How do I lower the pH in my soap?
Using additives such as powdered goat’s milk, flaxseed oil, olive oil, or sunflower oil will also help lower the pH level. But, the most important magic tip is to add citric acid. It’s one trick you can use from commercial soap makers. It is more effective if you add it after boiling lye and oil together.
Is shampoo base or acid?
Shampoo is considered acidic. Note: There were different types of shampoos available. But the main ingredient in shampoo is citric acid.
Is hand soap an acid?
Soap is a combination of a weak acid (fatty acids) and a strong base (lye), which results in what is known as “alkalai salt,” or a salt that is basic on the pH scale.
Can I add lemon juice to my soap?
I don’t recommend adding lemon juice to a melt and pour soap. The base can only accept so much liquid, especially, non-oil types. Lemon essential oil is best. Zest as I’ve used in this recipe is also fine, as it is mostly solid with some oils in the rind.
Can I add sugar to my black soap?
Different oils give different amounts and different types of lathers, so many soap makers turn to sugar to increase the suds. Adding a bit of sugar to a soap recipe can help make a light, bubbly lather with large bubbles when the oils you’re using do not lather up as much as you’d like.
Is baking soda a base or acid?
Its superpowers come from a two-letter term: pH. That stands for “power of hydrogen” to make something either an acid or a base (alkaline). Baking soda is an alkaline substance.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
