Are proteins polar or nonpolar?
Since proteins have nonpolar side chains their reaction in a watery environment is similar to that of oil in water. The nonpolar side chains are pushed to the interior of the protein allowing them to avoid water molecule and giving the protein a globular shape.
Are proteins non polar or polar?
2.2 Proteins
Ordinarily, proteins have a folded compact structure, in which nonpolar amino acids residues are located in the interior of molecular and hydrophilic residues located on the molecular surface.
What makes a protein polar?
All polar amino acids have either an OH or NH2 group (when in aqueous environment), and can therefore make hydrogen bonds with other suitable groups. Polar amino acid residues have a tendency to be on the outside of a protein, due to the hydrophilic properties of the side chain.
There are 10 non-polar amino acids found in protein core, and there are 10 polar amino acids. The essential amino acids in humans are: histidine, leucine, isoleucine, lysine, methionine, valine, phenylalanine, tyrosine and tryptophan.
What are non polar proteins?
Nonpolar amino acids include alanine (Ala), leucine (Leu), isoleucine (Ile), proline (Pro), tryptophan (Trp), valine (Val), phenylalanine (Phe), and methionine (Met). The side chains of these amino acids are long carbon chains or carbon rings, making them bulky. They are hydrophobic, meaning they repel water.
Proteins, made up of amino acids, are used for many different purposes in the cell. The cell is an aqueous (water-filled) environment. Some amino acids have polar (hydrophilic) side chains while others have non-polar (hydrophobic) side chains.
Are proteins water soluble?
The solubility of a protein in water depends on the 3D shape of it. Usually globular proteins are soluble, while fibrous ones are not. Denaturation changes the 3D structure so the protein is not globular any more. This has to do with the properties of the amino acids in the protein.
Is amino group polar or nonpolar?
An organic compound that contains an amino group is called an amine. Like oxygen, nitrogen is also more electronegative than both carbon and hydrogen, which results in the amino group displaying some polar character.
Amino acids are ordered from the most hydrophobic one, Isoleucine (I, on the left hand side) to the most hydrophilic one, Arginine (R, on the right hand side), according to the Kyte-Doolitle scale [2].
Are hydrophobic amino acids nonpolar?
Non-Polar Hydrophobic Amino Acids. Hydrophobic amino acids have little or no polarity in their side chains. The lack of polarity means they have no way to interact with highly polar water molecules, making them water fearing.
Why are amino acids non-polar?
Non-polar amino acids are a class of amino acids in which the variable R-group is comprised of mostly hydrocarbons; the amino acids cysteine and methionine also feature a sulphur atom, but (due to its similar negativity to carbon) this does not confer any polar properties to either of these amino acids.
Are lipids nonpolar or polar?
Lipids, i.e., fatty molecules, on the other hand, are non-polar, meaning that the charge distribution is evenly distributed, and the molecules do not have positive and negatively charged ends..
Hydrophobic Amino Acids
The nine amino acids that have hydrophobic side chains are glycine (Gly), alanine (Ala), valine (Val), leucine (Leu), isoleucine (Ile), proline (Pro), phenylalanine (Phe), methionine (Met), and tryptophan (Trp).
Which list of amino acids is considered nonpolar?
The non-polar amino acids (shown here) include: alanine, cysteine, glycine, isoleucine, leucine, methionine, phenylalanine, proline, tryptophan, tyrosine and valine.
Where are non-polar amino acids?
Non-polar amino acids tend to be found in the centre of the molecule (stabilise the structure) Polar amino acids tend to be located on the protein surface (capable of interacting with water molecules)
Why are proteins hydrophobic?
Structures of water-soluble proteins have a hydrophobic core in which side chains are buried from water, which stabilizes the folded state. Charged and polar side chains are situated on the solvent-exposed surface where they interact with surrounding water molecules.
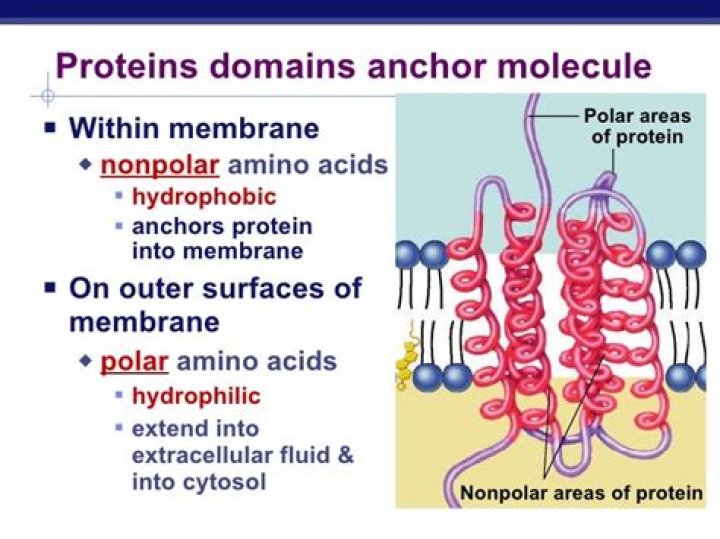
Most integral proteins contain residues with hydrophobic side chains that interact with fatty acyl groups of the membrane phospholipids, thus anchoring the protein to the membrane. Most integral proteins span the entire phospholipid bilayer.
Are protein channels hydrophilic?
Channel proteins have hydrophilic domains exposed to the intracellular and extracellular fluids; they additionally have a hydrophilic channel through their core that provides a hydrated opening through the membrane layers.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
