biological oxidation reduction, check these out | What is meant by biological oxidation?
Biological oxidation-reduction reactions, or simply biological oxidations utilize multiple stages or processes of oxidation to produce large amounts of Gibbs energy, which is used to synthesize the energy unit called adenosine triphosphate or ATP.
What is meant by biological oxidation?
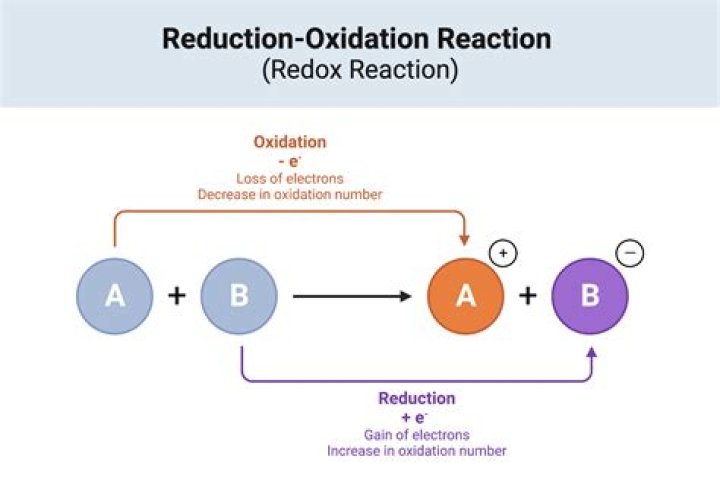
Biological oxidation is an energy-producing reaction in living cells, and it is coupled with a reduction reaction (Fig. 1). When a compound loses an electron, or is oxidized, another compound gains the electron, or is reduced. Oxidation-reduction (redox) reactions represent the main source of biological energy.
What is biological reduction?
Reduction generally means a reaction in which electrons are added to a compound; the compound that gains electrons is said to be reduced.
Why are oxidation reductions important in biology?
Oxidation-reduction (redox) reactions are important because they are the principal sources of energy on this planet, both natural or biological and artificial. Oxidation of molecules by removal of hydrogen or combination with oxygen normally liberates large quantities of energy.
What is oxidation and reduction bioenergetics?
oxidation: a reaction in which an element’s atoms lose electrons and its oxidation state increases. reduction: a reaction in which electrons are gained, and oxidation state is reduced, often by the removal of oxygen or the addition of hydrogen.
What is reduction biochemistry?
Reduction involves a half-reaction in which a chemical species decreases its oxidation number, usually by gaining electrons. The other half of the reaction involves oxidation, in which electrons are lost. Together, reduction and oxidation form redox reactions (reduction-oxidation = redox).
What is an example of an oxidation-reduction reaction?
The tarnishing of silver is just one example of a broad class of oxidation-reduction reactions that fall under the general heading of corrosion. Another example is the series of reactions that occur when iron or steel rusts. When heated, iron reacts with oxygen to form a mixture of iron(II) and iron(III) oxides.
What is meant by oxidation and reduction?
Oxidation and Reduction with respect to Oxygen Transfer. Oxidation is the gain of oxygen. Reduction is the loss of oxygen.
Are the cause of biological reduction?
Answer: pollution are the cause of biological reduction.
What are reducing agents in biology?
Reducing Agents, sometimes referred to as reductants or reducers, are elements or compounds that lose an electron to some other chemical species in a redox chemical reaction. A reducing agent is usually in one of its lower oxidation states and is known as the electron donor.
Why is reduction the term used to describe?
Why is reduction the term used to describe the gain of an electron? The electron acceptor’s net charge decreases. Which of the following statements regarding redox reactions is true? Redox reactions involve an oxidation reaction coupled with a reduction reaction. In metabolism, energy that is not used.
What is the role of oxidation and reduction?
Oxidation/reduction reactions
Oxidation refers to the loss of electrons by a substance. Oxygen may or may not be involved in the reaction. The substance oxidized is transformed into a second substance with a lower level of potential energy. Conversely, reduction refers to the acceptance of electrons by a substance.
Is NAD+ oxidized or reduced?
The cofactor is, therefore, found in two forms in cells: NAD+ is an oxidizing agent – it accepts electrons from other molecules and becomes reduced. This reaction, also with H+, forms NADH, which can then be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD.
What are the enzymes involved in biological oxidation?
Enzymes involved in oxidation and reduction are called oxidoreductases and are classified into four groups: oxidases, dehydrogenases, hydroperoxidases, and oxygenases. Oxidases use oxygen as a hydrogen acceptor.
What is reduction in metabolism?
Energy in metabolism often flows in terms of electrons. If electrons are lost, this is called oxidation. If electrons are gained, this is called reduction. Oxidation is coupled to reduction; that is, if something gets oxidized, then something else gets reduced.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
