dalton’s atomic theory definition, check these out | What are the 5 points of Dalton’s atomic theory?
/ (ˈdɔːltənz) / noun. chem the theory that matter consists of indivisible particles called atoms and that atoms of a given element are all identical and can neither be created nor destroyed. Compounds are formed by combination of atoms in simple ratios to give compound atoms (molecules).
What are the 5 points of Dalton’s atomic theory?
His theory contained five main propositions:
All matter is comprised of tiny, definite particles called atoms.Atoms are indivisible and indestructible.All atoms of a particular element share identical properties, including weight.Atoms of different elements contain different mass.
When was John Dalton’s atomic theory?
In 1808 John Dalton published his first general account of chemical atomic theory, a cornerstone of modern chemistry. The theory originated in his earlier studies of the properties of atmospheric gases.
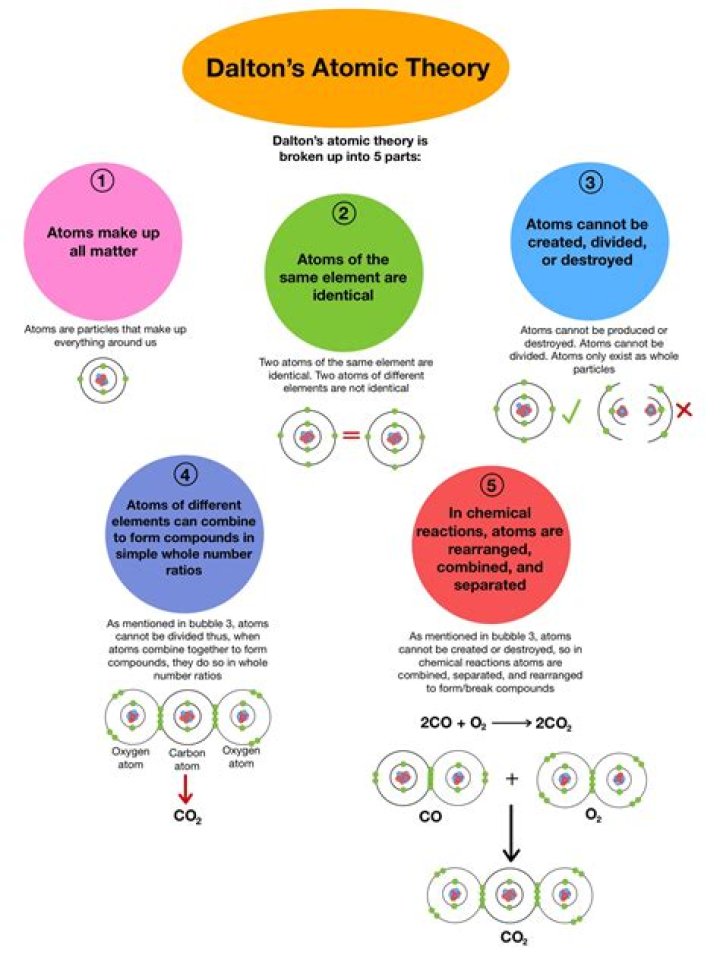
What are the 6 ideas of Dalton’s atomic theory?
Dalton’s Atomic Theory
All atoms of an element are identical. The atoms of different elements vary in size and mass. Compounds are produced through different whole-number combinations of atoms. A chemical reaction results in the rearrangement of atoms in the reactant and product compounds.
What are the 4 major ideas of Dalton’s atomic theory?
A theory of chemical combination, first stated by John Dalton in 1803. It involves the following postulates: (1) Elements consist of indivisible small particles (atoms). (2) All atoms of the same element are identical; different elements have different types of atom. (3) Atoms can neither be created nor destroyed.
Why is Dalton’s Atomic Theory important?
One of the most important merits of Dalton’s atomic theory is the fact that the theory does not violate several fundamental laws of chemical combination such as the law of definite proportions, the law of multiple proportions, and the law of conservation of mass.
What did Dalton’s atomic theory contribute?
In A New System of Chemical Philosophy, Dalton introduced his belief that atoms of different elements could be universally distinguished based on their varying atomic weights. In so doing, he became the first scientist to explain the behavior of atoms in terms of the measurement of weight.
What are the main ideas in Dalton’s Atomic Theory check all that apply?
What are the main ideas in Dalton’s atomic theory? Atoms combine in simple, whole number ratios to form compounds. All atoms of a given element have the same mass and other properties that distinguish them from the atoms of other elements. Atoms of one element cannot change into atoms of another element.
What are the main ideas in Dalton’s atomic theory How do they help explain the laws of conservation of mass of constant composition and of definite proportions?
The main idea for the dalton atomic theory is that everything is composed of atoms and atoms are called building blocks of matter.
How does Dalton’s atomic theory explain the law of multiple proportions?
The law, which was based on Dalton’s observations of the reactions of atmospheric gases, states that when elements form compounds, the proportions of the elements in those chemical compounds can be expressed in small whole number ratios.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
