Does aluminum lose or gain electrons?
Aluminum is a metal that will always lose three electrons. The halogens all have seven valence electrons. Each one of these elements wants to gain one electron to achieve an octet. Metals will always lose electrons to form cations.
Why does aluminum lose 3 electrons?
Initially, the aluminum atom had a charge of +13 + (−13) = 0; in other words, its charge was neutral due to the equal numbers of protons and electrons. When it becomes an ion, it loses 3 electrons, leaving behind only 10.
Does aluminum gain or loss?
An aluminum atom will lose up to three electrons when it forms an ion, creating the Al+, AL2+ or Al3+ cation.
The charge of an aluminum ion is typically 3+. This is because the element’s atomic number is 13, reflecting the fact that it has 13 electrons and 13 protons. The valence shell of aluminum has three electrons, and per the octet rule, these three electrons are lost resulting in just 10 electrons and 13 protons.
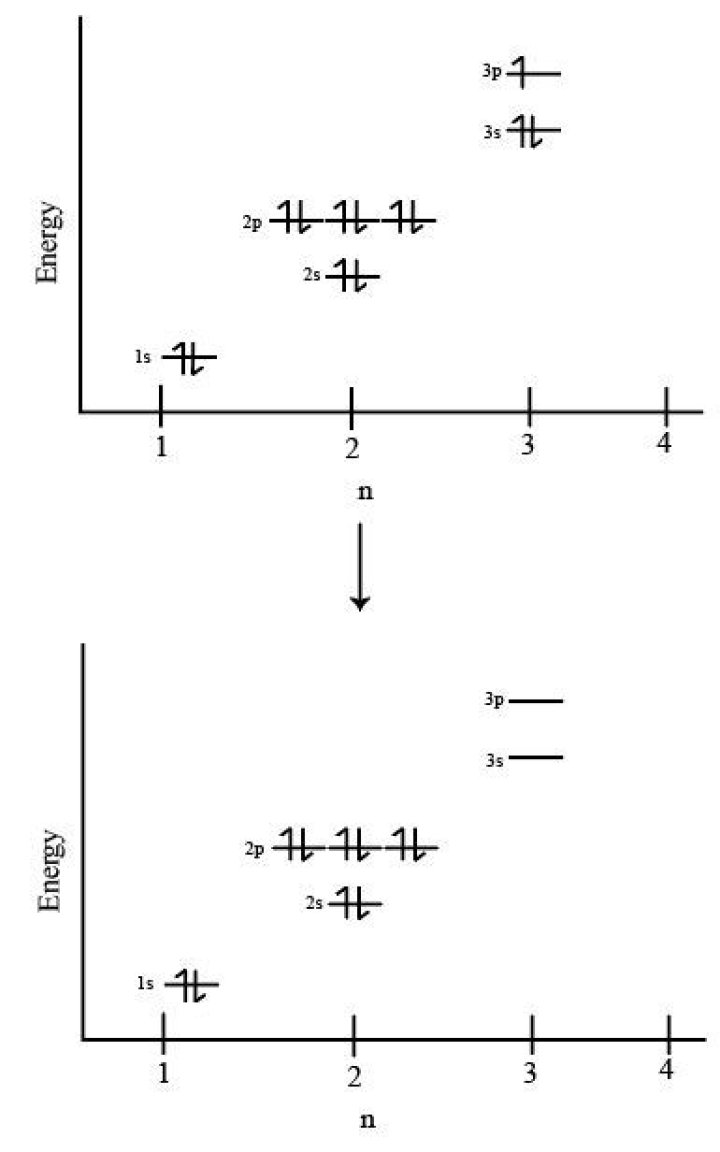
When aluminum loses electrons to form an aluminum ion electrons are lost from which Subshells?
When aluminum loses electrons to form an aluminum ion electrons are lost from which Subshell? Chloride ion (atomic number 17) has now got 17 protons but 18 electrons. This accounts for the -1 electrical charge. Aluminium atoms lose 3 electrons from the valence shell (3rd shell) to form an aluminium ion of +3 charge.
Oxygen is therefore also a really good oxidizing agent, and makes other elements lose electrons, while it gets reduced (gain electrons) itself. Reduction on the other hand, is opposite of oxidation, and is when the atom gains electrons, or loses an oxygen atom.
Why does aluminum have 10 electrons?
Also your teacher may ask you how many electrons are in Al^+3, the ionic form of aluminum, the +3 means your aluminum atom has a positive charge of 3 which means it has three less electrons than a nonionic atom of aluminum. so your answer will be 10 electrons in that case.
What does aluminum do to become stable?
An aluminum atom has three valence electrons in the third energy level 3s and 3p sublevels. Most atoms require 8 valence electrons in order to be stable. Metal atoms that have 3 or fewer valence electrons tend to lose electrons to form cations.
Boron and aluminum, from Group III (or 13), display different bonding behavior than previously discussed. These atoms each have three valence electrons, so we would predict that these atoms want to bond covalently in order to gain 5 electrons (through sharing) to fulfill the octet rule.
How does an Aluminium ion form?
Answer: Cations are the positive ions formed by the loss of one or more electrons. The aluminum atom loses its three valence electrons. The Mg 2+ ion, the Al3+ ion, the Na + ion, and the Ne atom are all isoelectronic.
What happens when an atom loses 3 electrons?
Answer: Atoms of elements can lose or gain electrons making them no longer neutral, they become charged. When an atom loses electron(s) it will lose some of its negative charge and so becomes positively charged. A positive ion is formed where an atom has more protons than electrons.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
