Does an electron have a mass of 1 AMU?
Protons, neutrons, and electrons: Both protons and neutrons have a mass of 1 amu and are found in the nucleus. … Electrons have a mass of approximately 0 amu, orbit the nucleus, and have a charge of -1.
What has a mass of 1 amu?
Since the nucleus accounts for nearly all of the mass of the atom, a single proton or single neutron has a mass of approximately 1 amu.
Is an electron mass 1?
The electron has a mass that is approximately 1/1836 that of the proton. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a longer de Broglie wavelength for a given energy.
The rest mass of the electron is 9.1093837015 × 10−31 kg, which is only 1/1,836the mass of a proton. An electron is therefore considered nearly massless in comparison with a proton or a neutron, and the electron mass is not included in calculating the mass number of an atom.
What’s the Amu of an electron?
Electrons have a mass of approximately 0 amu, orbit the nucleus, and have a charge of−1.
mp = mass of a proton (1.007277 amu) mn = mass of a neutron (1.008665 amu) me = mass of an electron (0.000548597 amu)
Which of the following has a charge of +1 and a mass of 1 amu?
Protons have a charge of +1, and a mass of approximately 1 atomic mass unit (amu).
Why is the mass of 1 amu slightly less than that of either a proton or neutron?
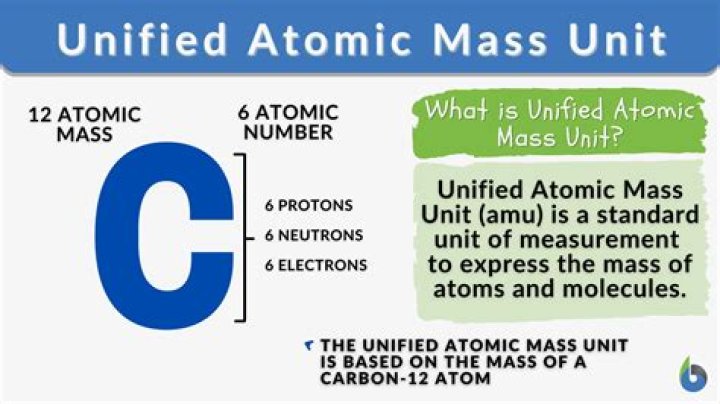
Since the mass of protons and neutrons are both 1 AMU, the mass of the atom expressed in AMU is the number of protons plus the number of neutrons. Electrons are much smaller than protons and neutrons, having a mass of approximately 0.0005 AMU, so their effect on the mass of the atom is negligible.
proton, stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10−27 kg, which is 1,836 times the mass of an electron.
What has a mass of 1 and an atomic number of 1?
Hydrogen, at the upper left of the table, has an atomic number of 1. Every hydrogen atom has one proton in its nucleus. Next on the table is helium, whose atoms have two protons in the nucleus.
What is a neutrons mass in amu?
Mass of neutron = 1.008665 amu = 1.67495*10-27 kg. Mass of proton = 1.007277 amu = 1.6726*10-27 kg. Amu stands for atomic mass unit. Obviously, the mass of a proton and neutron are very similar, while the mass of an electron is much less than the other two particles.
What does a +1 charge mean?
If the number of assigned electrons is less than the Group Number, the the Formal Charge is the difference between the Group Number and the number of assigned electrons (e.g., if assigned number of electrons is 4 and the atom is nitrogen with a Group Number of 5 (Group V), then the Formal Charge would be +1, meaning it
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
