Does ethyl-4-aminobenzoate react with HCl?
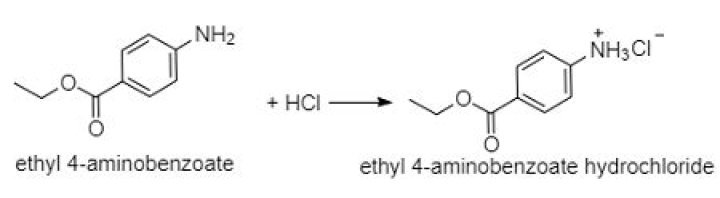
When ethyl-4-aminobenzoate reacts with hydrochloric acid, the amine hydrochloride salt of ethyl-4-aminobenzoate is formed. i.e. Because it is an ionic salt, ethyl-4-aminobenzoate hydrochloride is far more soluble than ethyl-4-aminobenzoate itself.
Is 4 ethyl aminobenzoate an acid or base?
Naphthalene is neutral, benzoic acid is acidic, and ethyl 4-aminobenzoate is basic.
Is aminobenzoate an acid or base?
4-aminobenzoate is an aromatic amino-acid anion that is the conjugate base of 4-aminobenzoic acid. It has a role as an Escherichia coli metabolite, a plant metabolite and a Saccharomyces cerevisiae metabolite. It is an aromatic amino-acid anion and an aminobenzoate.
Is ethyl-4-aminobenzoate polar or nonpolar?
The ethyl-4-aminobenzoate is similar to benzoic acid, in that it has a nonpolar ring and has polar attachments. The nonpolar ring is what makes it soluble in diethyl ether.
Is NaOH a base?
Now let’s look at lye, a strong base with the chemical formula NaOH (sodium hydroxide).
Is ethyl p aminobenzoate an acid?
Benzocaine is a benzoate ester having 4-aminobenzoic acid as the acid component and ethanol as the alcohol component.
Is benzoic acid soluble in Naoh?
For example, benzoic acid is not soluble in water, yet it is soluble in sodium hydroxide solution and in sodium hydrogen carbonate solution because these bases react with benzoic acid to form the water-soluble benzoate ion.
What is benzocaine soluble in?
Benzocaine occurs in two forms: a crystalline salt (benzocaine HCl) that is soluble in water at 0.4 g/L, and a nonwater-soluble basic form that must be dissolved in ethyl alcohol at a concentration of 0.2 g/ml before it is sufficiently soluble in water to create a functional induction dose.
What is the solubility of benzoic acid?
Benzoic acid or benzene-carbonic-acid is a monobasic aromatic acid, moderately strong, white crystalline powder, very soluble in alcohol, ether, and benzene, but poorly soluble in water (0.3 g of benzoic acid in 100 g of water at 20 °C).
What is PABA made of?
It’s found in brewer’s yeast, organ meat, mushrooms, whole grains, and spinach (1, 2 ). Your body can also synthesize the compound using certain bacteria in your gut. PABA then assists in the production of folate (vitamin B9).
What is PABA good for?
PABA is also used to treat infertility in women, arthritis, “tired blood” (anemia), rheumatic fever, constipation, systemic lupus erythematosus (SLE), and headaches. It is also used to darken gray hair, prevent hair loss, make skin look younger, and prevent sunburn.
What is PABA used for?
PABA is taken for a disease that causes curved, painful erections (Peyronie disease), a rare autoimmune disorder that causes skinblisters (pemphigus), hardening of skin and connective tissue (scleroderma), and many other conditions.
Is NaOH polar or nonpolar?
In alkali metal hydroxides, such as NaOH, the Na-O bond is most polar. The electronegativity difference between sodium and oxygen is larger than that between oxygen and hydrogen.
Is HCl polar or nonpolar?
Is HCl polar or nonpolar? Because chlorine is more electronegative than hydrogen, hydrochloric acid HCl forms a polar bond, and is therefore a polar molecule. There is no symmetry which could cancel out the dipole charge.
What is the pKa of benzocaine?
The pKa of benzoic acid is 4.2; the pKa of the conjugate acid of benzocaine is 2.5.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
