Does KCl contain ionic bonds?
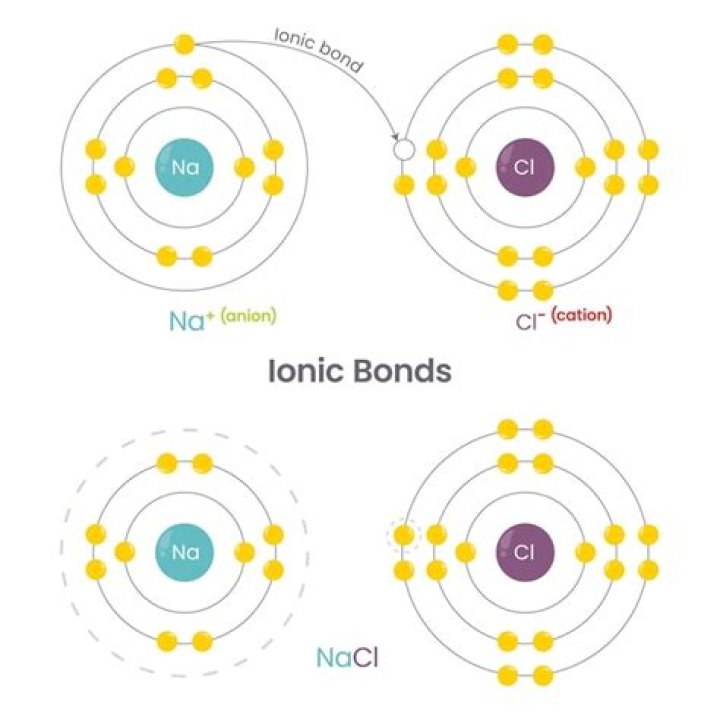
Yes, KCl, or potassium chloride, is an ionic bond.
Is KCl an ionic compound yes or no?
Potassium chloride is an ionic compound and is a salt that naturally occurs as a solid that has a powdery and crystalline appearance. Its chemical formula is KCl: it consists of one potassium (K+) ion and one chlorine (Cl-) ion.
What type of bond is KCl?
So, is KCl ionic or covalent? Yes, KCl is ionic in nature as the electronegativity of Potassium is 0.82 while that of Chlorine is 3.16 and hence, the difference between the electronegativity of these two elements is 2.34 which is greater than 2.0, required for the formation of ionic bond.
Silver sulfide is a network solid made up of silver (electronegativity of 1.98) and sulfur (electronegativity of 2.58) where the bonds have low ionic character (approximately 10%).
How is the bonding in KCl formed?
A positive ion, K+ is formed. The oppositely-charged ions formed, K+ and Cl–, are then strongly attracted to each other by strong electrostatic forces in the crystal lattice, called ionic bonds or electrovalent bonds. Hence, the ionic compound potassium chloride with the formula KCl is formed.
KCl is more ionic because the ionic character increases as down the group while decreases along the period in the periodic table. Also, the bonding in KCl is more ionic than in NaCl. Electronegativity scales also support that NaCl is less ionic when compared to KCl.
Is CaS molecular or ionic?
Calcium sulfide, CaS , exhibits an ionic bond.
Is H2 ionic or covalent?
Covalent molecules made of only one type of atom, like hydrogen gas (H2), are nonpolar because the hydrogen atoms share their electrons equally.
As we can see that there is loss and gain of electrons between chlorine and magnesium so magnesium chloride is an ionic compound and there is electrostatic attraction between them so it is an electrovalent compound also.
Is CA NO3 2 an ionic compound?
The name of the compound Ca(NO3)2 is Calcium nitrate. An ionic compound, is a compound that contains a metal element in its formula unit. Metal elements are found to the left of the stairs in a periodic table ( refer to the periodic table at the end of this answer ).
Is fe2o3 an ionic compound?
It is represented by Fe2O3. The oxidation state of iron oxide is +3 and +2. The bond formed between iron and oxygen is due to the difference in electronegativity between the two atoms. Since iron is metal and oxygen is non-metal therefore the bonding between oxygen and iron is ionic.
Is KCl neutral basic or acidic?
-The ions from $KCl$ derived from a strong acid (HCl) and a strong base (KOH). Therefore, neither ion will affect the acidity of the solution so, $KCl$ is a neutral salt.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
