Does NO2 have linear shape?
Why are NO2 and CO2 different shapes? two N=O double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2.
What shape does NO2 have?
We can see that NO2 has a bent molecular geometry and the angle is around 120 degrees.
Is NO2 linear and polar?
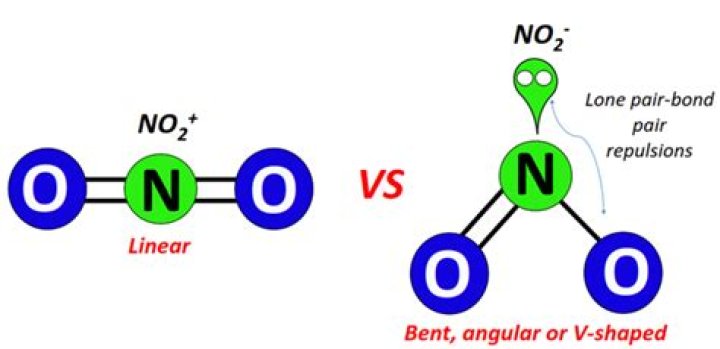
Answer: NO2+ is a nonpolar molecule because of its linear structure which cancels out opposing dipole forces. Answer: NO2 is polar molecule due to the presence of a lone pair of electrons whose electron-electron repulsion results in a bent structure.
2 Answers. Nitrous oxide is linear.
Is NH3 linear?
For example; four electron pairs are distributed in a tetrahedral shape. If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4). If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3).
Why are NO2 and CO2 different shapes? two N=O double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2. a greater repulsion than the single electron in NO2, so the O-N-O angle is reduced further, to 115.4°.
Is NO2 a structure?
Nitrogen Dioxide (NO2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. At room temperatures, nitrogen dioxide is a reddish-brown gas that has a density of 1.8 g/dm3.
Is NO2 bent or trigonal planar?
However, the shape of the molecule is described in terms of the positions of the atoms, not the lone electron pairs. Thus, NO2– is said to have a bent shape, not a trigonal planar one. It is assumed that the lone pair is “invisible” when describing the shape of the molecular geometry.
It is isoelectronic with CO2, having two N=O. double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2.
Why is NO2 trigonal planar?
The nitrogen and and one oxygen are bonded through a double bond which counts as “one electron pair”. Hence the molecule has three electron pairs and is trigonal planar for electron pair geometry. Nitrogen dioxide is a main component of photochemical smog air pollution.
Is BrO3 trigonal planar?
The total valence electron available for drawing the Bromate ion (BrO3-) lewis structure is 26. The hybridization in BrO3- is Sp3. Total 16 lone pairs electrons and 10 bonded pairs electrons present in BrO3- lewis structure. The molecular geometry of BrO3- is trigonal pyramidal and electron geometry is tetrahedral.
Does NO2 have polar or nonpolar bonds?
NO2+ (Nitronium ion) is nonpolar in nature because it has a linear geometrical structure due to which polarity of opposite NO bonds gets canceled by each other resulting in the nonpolar NO2+ ion. Nitronium ion is a stable ion in normal conditions.
NCl3 is a slightly polar molecule because of the small difference between the electronegativity of nitrogen and chlorine atom. NCl3 molecule has one lone pair that leads to repulsion between electrons and the shape of the molecule is trional pyramidal.
Is NH3 polar or nonpolar?
Yes, we can say that NH3 is a polar covalent bond. Nitrogen forms a covalent bond with three atoms to form a molecule. You can get the difference between the N-H bond and the NH3 compound since the two are polar, even in their gaseous state.
Why N2O is linear but polar?
We know that $ C{O_2} $ is a linear and symmetrical molecule so the net dipole moment is zero. Hence, it is non polar. Now, $ {N_2}O $ is also linear and symmetrical but it is considered as the resonance hybrid of two structures due to which its net dipole moment is $ 0.116D $ . Hence, it is polar.
Why is N2O linear?
In the diagram given above, the type of bonding in nitrous oxide is covalent bonding. Due to the presence of sigma bonds and absence of lone pairs on the central atom, the stable structure of nitrous acid is linear. It is also known as sulphur dioxide.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
