Does sand have a covalent or ionic bond?
Silica is the main compound found in sand. It is an example of a giant covalent substance. It contains many silicon and oxygen atoms. These are joined together by covalent bonds in a regular arrangement, forming a giant covalent structure .
Is sand an ionic or molecular compound?
Sand is a covalent compound. It did not dissolve in water and was not conductive. Additionally, its chemical formula, SiO2, shows that it is made of silicon and oxygen, two nonmetals. Since covalent compounds are made up of only nonmetals, sand is a covalent compound.
Is sand a covalent network solid?
Covalent network solids include crystals of diamond, silicon, some other nonmetals, and some covalent compounds such as silicon dioxide (sand) and silicon carbide (carborundum, the abrasive on sandpaper). The atoms in these solids are held together by a network of covalent bonds, as shown in Figure 5.
Silica is the main compound found in sand. It is an example of a giant covalent substance. It contains many silicon and oxygen atoms. These are joined together by covalent bonds in a regular arrangement, forming a giant covalent structure .
Is sand polar or nonpolar?
Sand is a silicate mineral with the formula SiO2. Sand grain surfaces are convalently bonded to hydrogen atoms which are polar bonds. Thus both water and sand contain polar molecules allowing water to be attracted to sand (remember “like with like”).
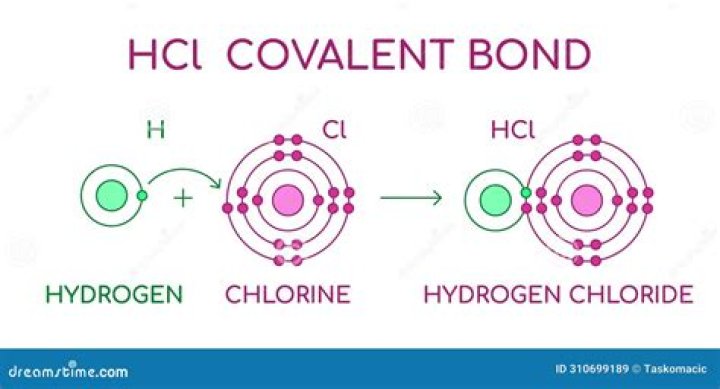
The hydrogen molecule is the simplest substance having a covalent bond. It forms from two hydrogen atoms, each with one electron in a 1s orbital. Both hydrogen atoms share the two electrons in the covalent bond, and each acquires a helium-like electron configuration. A similar bond forms in Cl2.
Is NaCl ionic or covalent?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
Does NaOH have ionic and covalent bonds?
Does NaOH contain both ionic and covalent bonds? Some atoms form polyatomic ions. In each hydroxide ion the Oxygen atom and the H atom are bonded via covalent bonds, but the OH- and Na+ are held together in a crystal lattice via ionic bonds, thus making NaOH an ionic compound.
CaCl2 is an ionic bond. This is because the calcium gives up an electron to each of the chlorine atoms resulting in the calcium becoming Ca2+ ions
Which of the following compound is covalent?
sol. H2 is a covalent, diatomic, molecule with a sigma covalent bond between two hydrogen atoms. I HOPE IT HELPFUL FOR YOU.
Is hydrogen iodide covalent or ionic?
Hydrogen iodide is a diatomic molecule formed by a covalent bond. The electronegativity of hydrogen is approximately 2.2 while iodine has an
What is a covalent bond and give examples?
By definition, a pure covalent bond is one that exists between two atoms with the same electronegativities. Thus, a pure covalent bond does not display any ionic character. Diatomic elements are perfect examples of pure covalent bonds because both the atoms evenly share the electrons. Examples: H2, O2, and N2.
Ionic bond examples include:
LiF – Lithium Fluoride.LiCl – Lithium Chloride.LiBr – Lithium Bromide.LiI – Lithium Iodide.NaF – Sodium Fluoride.NaCl – Sodium Chloride.NaBr – Sodium Bromide.NaI – Sodium Iodide.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
