Does zinc lose or gain electrons?
Zinc has a strong tendency to lose electrons. The Zn metal is more reactive than copper. In an acidic solution, Zn atoms lose electrons to H+ ions, but copper atoms will not. The tendency is measured in terms of standard reduction potential.
Does zinc want to gain or lose electrons?
The tendency of a metal to lose electrons depends on its reactivity. The more reactive a metal is, the easier it will lose electrons. The negative E∘ for zinc tells you that the equilibrium lies to the left, meaning which confirms that zinc is more reactive and will tend to lose electrons to form Zn2+ cations.
Will Zn gain electrons?
When a metal electrode is dipped in an electrolyte containing its own ion, then it may lose or gain electrons depending on whether it has a negative or a positive oxidation potential, again involving energy (W=qV). Thus, Zn having a negative potential loses electrons and Cu having a positive potential gains electrons.
So if zinc lost electrons, zinc is oxidised. Than means zinc is providing its electrons to copper, and copper is reducing because of that.
Why does zinc lose electrons to copper?
In a zinc-copper voltaic cell, it is the copper(II) ions that will be reduced to copper metal. That is because the Cu 2+ ions have a greater attraction for electrons than the Zn 2+ ions in the other half-cell. Instead, the zinc metal is oxidized.
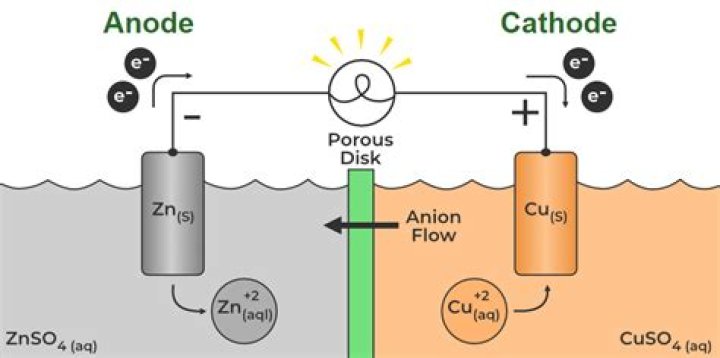
The zinc electrode becomes lighter as zinc atoms are oxidized to Zn2+ ions, which go into solution. The copper electrode becomes heavier as Cu2+ ions in the solution are reduced to copper metal. The concentration of Zn2+ ions at the anode increases and the concentration of the Cu2+ ions at the cathode decreases.
Why does zinc dissolve?
The solubility of zinc depends on temperature and pH of the water in question. When the pH is fairly neutral, zinc in water insoluble. Solubility increases with increasing acidity. Zinc dissolves in water as ZnOH+ (aq) or Zn2+ (aq).
Can zinc have +1 charge?
Zinc will rarely form ions with a +1 charge but it will never form ions with a negative charge. As zinc is a metal, it generally forms metallic compounds with other metals. Because it has an ion charge of +2, zinc ions are strong reducing agents and readily form ionic bonds.
The atomic number of zinc is 30 so, 30 electrons are there in the atom.
What reaction occurs when zinc ions gain electrons?
Gain of electrons is reduction. A redox reaction is one in which both oxidation and reduction are occurring simultaneously.
What happens to the electrons when the zinc is added to the copper?
Reduction occurs when the oxidation number of an atom becomes smaller. Oxidizing agents gain electrons while reducing agents lose electrons. In the first reaction, the copper ion is able to oxidize the zinc metal. However, in the second reaction, the zinc ion is not able to oxidize the copper metal.
What is the role of zinc in the cell?
Zinc is essential for cell proliferation and differentiation, especially for the regulation of DNA synthesis and mitosis. On the molecular level, it is a structural constituent of a great number of proteins, including enzymes of cellular signaling pathways and transcription factors.
The reducing agent is the one that reduces other elements thereby oxidizing on its own. So zinc will undergo oxidation and give up two electrons. The liberated two electrons from zinc will now be added to copper ions forming copper metal. Thus, zinc can replace copper from copper sulphate solution.
Why zinc is more reactive than copper?
The metallic bond of copper is therefore stronger than zinc, so more energy is required to break this bond in order for it to react. Since more energy is required for copper to react, we can say that zinc is more reactive than copper.
Does the zinc electrode gain lose or retain the same mass as the reaction proceeds?
Because Zn(s) is oxidized in the cell, the zinc electrode loses mass, and the concentration of the Zn2+ solution increases as the cell operates. Similarly, the Cu electrode gains mass, and the Cu2+ solution becomes less concentrated as Cu2+ is reduced to Cu(s).
Why zinc is anode in galvanic cell?
Zinc behaves as the anode (supplying electrons) of the galvanic cell and the copper as the cathode (consuming electrons). The zinc electrode is dissolved (corroded or oxidized) and the copper electrode accepts copper atoms from the electrolyte (electroplating or reduction).
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
