enzymatic reaction rate, check these out | What is the reaction rate of an enzyme?
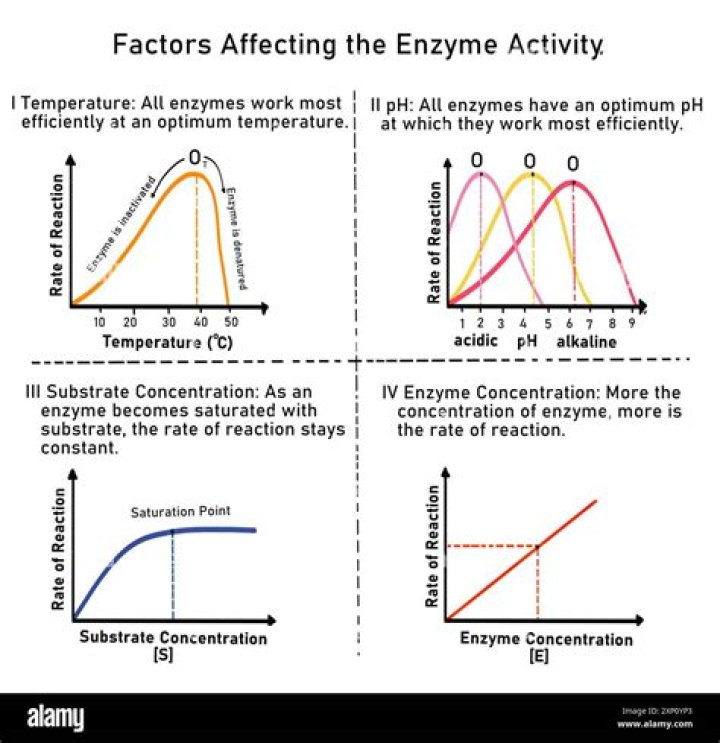
The rate of an enzyme-catalyzed reaction increases with an increase in the concentration of an enzyme. At low temperatures, an increase in temperature increases the rate of an enzyme-catalyzed reaction. At higher temperatures, the protein is denatured, and the rate of the reaction dramatically decreases.
What is the reaction rate of an enzyme?
Reaction rate is the speed at which the reaction proceeds toward equilibrium. For an enzyme catalyzed reaction, the rate is usually expressed in the amount of product produced per minute. Reaction rate is governed by the energy barrier between reactions and products.
How do you calculate the rate of an enzymatic reaction?
The rate of reaction can be calculated according to the following formula: Rate of reaction (s–1) = 1 / time taken (s)
How does enzyme increase the rate of reaction?
Enzymes (and other catalysts) act by reducing the activation energy, thereby increasing the rate of reaction. The increased rate is the same in both the forward and reverse directions, since both must pass through the same transition state.
What is the Lineweaver Burk equation?
The Lineweaver-Burk equation is a linear equation, where 1/V is a linear function of 1/[S] instead of V being a rational function of [S]. The Lineweaver-Burk equation can be readily represented graphically to determine the values of Km and Vmax.
What is the rate of reaction in biology?
The rate of a reaction is the speed at which a reaction occurs. It is the rate at which reactants are converted into products.
What is the formula for rate?
However, it’s easier to use a handy formula: rate equals distance divided by time: r = d/t.
How do u calculate rate?
Here are the steps for calculating rate:
Identify the measurements being compared. Compare the measurements side-by-side. Simplify your calculations by the greatest common factor. Express your found rate. Find the difference between the two data values. Divide the difference by the original number. Multiply the results by 100.
What is initial rate of enzymatic reaction?
The initial rate of reaction is when concentrations of enzyme and substrate are known, so this allows fair comparison if you then change initial concentrations of enzymes or substrate. Plotting reaction rate against substrate concentration typically gives a curve that is similar in shape to the product/time plot.
How does enzyme concentration affect the rate of reaction?
Enzyme concentration: Increasing enzyme concentration will speed up the reaction, as long as there is substrate available to bind to. Once all of the enzymes have bound, any substrate increase will have no effect on the rate of reaction, as the available enzymes will be saturated and working at their maximum rate.
What is the relationship between enzyme concentration and reaction rate?
The relationship between enzyme concentration and the rate of the reaction is usually a simple one. If we repeat the experiment just described, but add 10% more enzyme, the reaction will be 10% faster, and if we double the enzyme concentration the reaction will proceed twice as fast.
Why does higher enzyme concentration increased rate of reaction?
Increasing Substrate Concentration increases the rate of reaction. This is because more substrate molecules will be colliding with enzyme molecules, so more product will be formed.
What is Km and Vmax?
Vmax is the maximum velocity of the reaction at which all the enzymes get saturated with the substrate. Km is the concentration of substrate at which half of the maximum velocity is achieved.
What is an enzyme reaction?
An enzyme attracts substrates to its active site, catalyzes the chemical reaction by which products are formed, and then allows the products to dissociate (separate from the enzyme surface). The combination formed by an enzyme and its substrates is called the enzyme–substrate complex.
What may affect the rate of an enzyme driven?
Several factors affect the rate at which enzymatic reactions proceed – temperature, pH, enzyme concentration, substrate concentration, and the presence of any inhibitors or activators.
How do you calculate the absorbance rate of an enzyme reaction?
You need to correlate the absorbance of the product released in your assay with standard product curve. By using y=mx+c, from your (Standard curve) you need to check the concentration of product released in term of micro gram. After identifying the amount of product release, then you can calculate Enzyme activity.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
