how are metalloids designated in the periodic table, check these out | Where are metalloids designated in the periodic table?
Where are metalloids designated in the periodic table?
The periodic table on the left separates elements into three groups: the metals (green in the table), nonmetals (orange), and metalloids (blue).
How many metalloids are designated in periodic table?
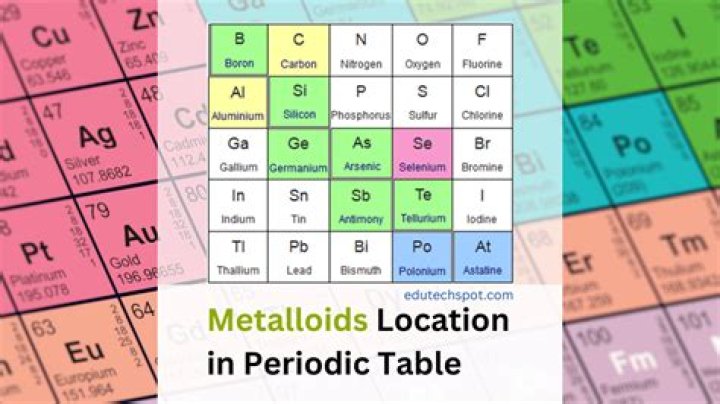
The six commonly recognised metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium. Five elements are less frequently so classified: carbon, aluminium, selenium, polonium, and astatine.
How are metals designated in periodic table?
The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. The semimetals lie along a diagonal line separating the metals and nonmetals.
What is group 1 on the periodic table called?
The alkali metals are six chemical elements in Group 1, the leftmost column in the periodic table. They are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).
How do metalloids compared to metals and nonmetals?
Metals, nonmetals and metalloids are elements that are found in the earth. The main difference between metals nonmetals and metalloids is that metals show the highest degree of metallic behavior and nonmetals do not show metallic behavior whereas metalloids show some degree of metallic behavior.
How are metalloids different from metals and nonmetals?
Metalloids have properties in between those of the metals and non-metals and are semiconductors (whereas metals are conductors and nonmetals are not conductors) and used extensively in circuitry (like computer chips). They can be shiny (have luster) or not whereas all metal are shiny.
What are the 7 metalloids in the periodic table?
Which Elements Are Metalloids?
Boron (B)Silicon (Si)Germanium (Ge)Arsenic (As)Antimony (Sb)Tellurium (Te)Polonium (Po)
How is the periodic table divided?
The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). Elements in the same group have the same number of valence electrons. Meanwhile, elements in the same period have the same number of occupied electron shells.
Which one of the following is metalloids?
The elements boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), polonium (Po) and astatine (At) are considered metalloids. Metalloids conduct heat and electricity intermediate between nonmetals and metals and they generally form oxides.
What metal is grouped with metalloids?
The post-transition metals cluster to the lower left of this line. Metalloids: The metalloids are boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te) and polonium (Po). They form the staircase that represents the gradual transition from metals to nonmetals.
Are metalloids ductile?
Malleability: Metalloids can be malleable and ductile, like metals. They also can be brittle, similar to nonmetals.
Are metalloids semiconductors?
A number of the metalloids are semiconductors as they have intermediate conductivity which is temperature dependent. The ability of the metalloids to conduct electricity and heat is far better than the nonmetals, for example, diamond , which are insulators.
What element is in period 4 Group 12?
zinc group element, any of the four chemical elements that constitute Group 12 (IIb) of the periodic table—namely, zinc (Zn), cadmium (Cd), mercury (Hg), and copernicium (Cn).
What is Group 2 called?
The Group 2 alkaline earth metals include Beryllium, Magnesium, Calcium, Barium, Strontium and Radium and are soft, silver metals that are less metallic in character than the Group 1 Alkali Metals.
What is the name of Group 17 on the periodic table?
halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
How are metalloids similar to metals?
Metalloids are similar to metals in that they both have valence orbitals that are highly delocalized over macroscopic volumes, which generally allows them to be electrical conductors.
How are metals nonmetals and metalloids arranged on the periodic table?
The metals are to the left of the line (except for hydrogen, which is a nonmetal), the nonmetals are to the right of the line, and the elements immediately adjacent to the line are the metalloids.
How are metals and metalloids similar and different?
A metal is a substance with high heat and high electrical conductivity. A metalloid has some features similar to metals and some features similar to nonmetals. Nonmetals have low conductivity of heat and electricity.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
