How do you calculate the pH of an aniline?
Hence, the pH of the solution is 7.818. The ionization constant of the conjugate acid of aniline is calculated by dividing the ionic product of water by the ionization constant of aniline.
How do you calculate the pH of an aniline?
pH lectures
Aniline pKb=9.4. Aniline in solution of its hydrochloride is in form of conjugate acid. To calculate pH of such solution we have to find pKa for aniline and treat it like weak acid. pKa=14-9.4=4.6 – and this value we will put into simplified formula 8.13.
What is the pH of a 0.1 M solution of aniline?
When 0.1 M, 50 mL solution of aniline was mixed with 0.1 M, 25 mL solution of HCl, the pH of the resulting solution was 8.
What is Ka of aniline?
Aniline hydrochloride, C_6H_5NH_3Cl, is a weak acid. Its conjugate base is the weak base aniline, C6H5NH2. Ka = 2.40times 0-5.
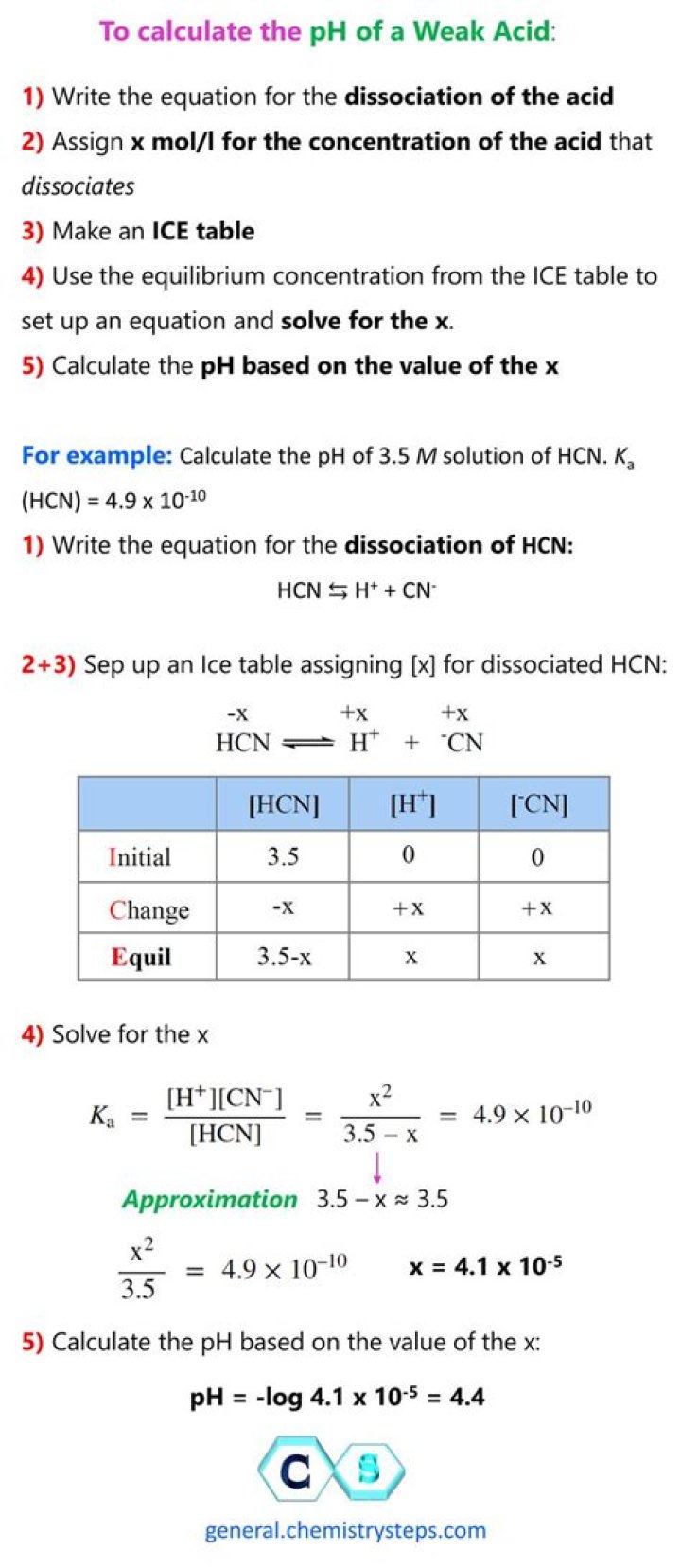
How do you calculate pH?
To calculate the pH of an aqueous solution you need to know the concentration of the hydronium ion in moles per liter (molarity). The pH is then calculated using the expression: pH = – log [H3O+].
What is the pH of pyridine?
Therefore it determines pH of a 0.2M solution of pyridine is 9.24.
What’s the pH of ammonia?
Ammonia: pH 11-13.
What is OH ion concentration is 0.1 M solution of aniline?
5×10−11M.
What is the pKb value of aniline?
The pKb values of ammonia, methenamine and benzenamine (aniline) are 4.75, 3.38 & 9.38 respectively.
What is the kb of aniline?
Now, We will find kb expression of aniline (C6H5NH2), the kb expression is, C6H5NH2 (aq) + H2O (l) – C6H5NH3 +(aq) + oH – (aq).
Is C6H5NH3 an acid?
Therefore, Cl- does NOT spontaneously pick up an H+ to form HCl in solution. C6H5NH3+ now looks like a derivative of the ammonium ion! This is a weak acid.
Is C6H5NH3 a weak acid?
Aniline hydrochloride, (C6H5NH3)Cl, is a weak acid. Relevance. The acid can be titrated with a strong base such as NaOH.
Is C6H5NH2 an acid or base?
Aniline, C6H5NH2(l), is an aromatic organic compound that is a base.
How do you calculate pH from KW?
Determining pH and Kw
We can determine the hydrogen ion concentration in water from measuring its pH.Pure water has pH = 7. That means the [H+1] = 1.0 x 10-7 M.Because you get one OH-1 for each H+1 the [OH-1] = 1.0 x 10-7 M.Therefore Kw = [H+1][OH-1] = 1.0 x 10-14 M.
How do you find pH from KB?
The Henderson-Hasselbalch equation states that pOH = pKb + log([salt]/[base]). Hence, assuming you know the values of [salt] and [base], you can take the negative log of Kb. Adding pKb and log([salt]/[base]) will then give you your pOH value. From there, subtract pOH from 14 to find pH as pH + pOH = 14.
Can pH be negative?
It’s definitely possible to calculate a negative pH value. In practice, any acid that yields a concentration of hydrogen ions with a molarity greater than 1 will be calculated to have a negative pH. For example, the pH of 12M HCl (hydrochloric acid) is calculated to be -log(12) = -1.08.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
