How do you know if its paramagnetic or diamagnetic?
Why is Au diamagnetic? – Quora. i think u are about to write Au+ , taking that into consideration i am answering the question . since , all orbitals are filled with electrons , then the ion is diamagnetic , in reverse case it will be paramagnetic .
How do you know if its paramagnetic or diamagnetic?
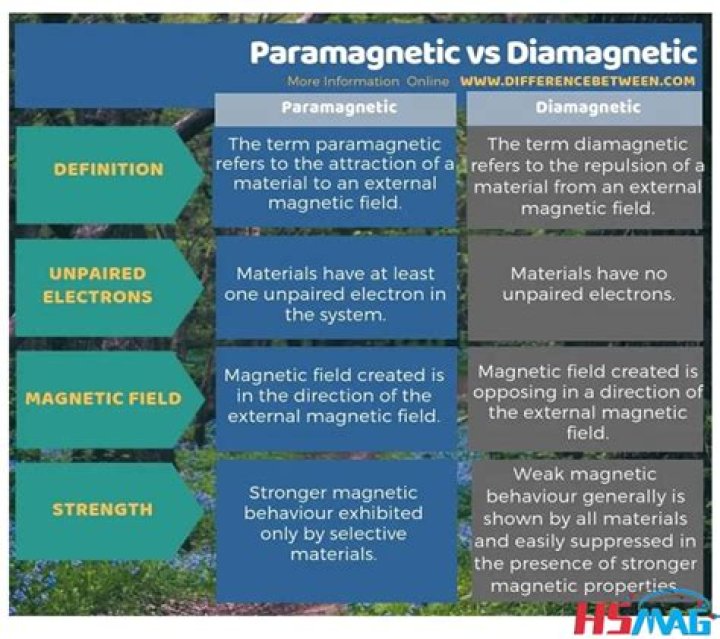
The magnetic properties of a substance can be determined by examining its electron configuration: If it has unpaired electrons, then the substance is paramagnetic and if all electrons are paired, the substance is then diamagnetic.
Is Cd2 ++ paramagnetic or diamagnetic?
All electrons are paired, making the neutral molecule Cd2 diamagnetic. Hence, Cd+2 , with one less electron from a fully-occupied orbital, is paramagnetic.
Is Mo6+ paramagnetic or diamagnetic?
Dilution of the Mn3+ magnetic array by the paramagnetic (Cu2+) and diamagnetic (Mo6+) cations is found to decrease the antiferromagnetic ordering temperature and it becomes undetectable for x ⩾ 0.
Which species are paramagnetic?
Paramagnetic materials include aluminium, oxygen, titanium, and iron oxide (FeO).
Which ions is paramagnetic?
Paramagnetic is a magnetic state of an atom with one or more unpaired electrons. These unpaired electrons get attracted by the magnetic field due to the magnetic dipole moments of the electrons. Diatomic oxygen is an example of a paramagnetism. Cr3+ with three unpaired electrons is an example of paramagnetic ions.
Is V3+ paramagnetic or diamagnetic?
V3+ (Vanadium 3+ ion) is paramagnetic since it has two unpaired electrons and therefore, it can get easily magnetised in presence of the external magnetic field.
Which of the following is a diamagnetic ion v2+ Sc3+ cu2+ mn3+?
The correct answer is – V²⁺ and Sc³⁺. Diamagnetic materials are compounds that are repelled by a magnetic field.
What species is diamagnetic?
Diamagnetic species are atoms that has zero unpaired electrons. We can determine this by setting up the orbital diagrams of the valence electrons for each atom.
Is carbon a paramagnetic?
We have unpaired electrons here for carbon when we draw out the orbital notation. And unpaired electrons means that carbon is paramagnetic. So carbon is paramagnetic.
Is Ca2+ paramagnetic or diamagnetic?
Ca2+ is believed to be paramagnetic due to the excitation of one electron from the s-orbital to the emptied d-orbital (s and d orbital are closer in energy, thereby causing transition to occur between both orbitals) which renders the s orbital unpaired in its excited state and attracted to the magnetic field (PAULI’ s
Is carbon a diamagnetic molecule?
Solution: Carbon molecule (C₂) with 12e⁻ is diamagnetic because all the electrons are paired. Though O₂ has 16e⁻ but it is paramagnetic due to the presence of two unpaired e⁻ in the antibonding πMOs. Superoxide ion (O₂⁻) is also paramagnetic as it has one unpaired e⁻.
Is Y3+ paramagnetic or diamagnetic?
Y3+ ion is a type of ion that is diamagnetic in nature. This element has an atomic number 39.
Is phosphorus diamagnetic or paramagnetic?
there are 3 unpaired electrons in the 2p orbital. Phosphorus (P) is diamagnetic.
Is Ti4+ paramagnetic or diamagnetic?
Ti4+ has no unpaired electrons and is diamagnetic.
Which of the following species is not paramagnetic?
Thus, only CO does not fulfil the condition of paramagnetic character that is the compound must have unpaired electrons. So it is not a paramagnetic species. Therefore, the correct option for this given question is A that is carbon monoxide (CO).
Which one of the following is a paramagnetic?
Paramagnetic species contains unpaired electrons in their molecular orbital electronic configuration. Thus, among the given species only O-2 is paramagnetic.
IS NO+ a paramagnetic species?
Explanation: CN- , CO and NO+ are isoelectronic with 14 electrons each and there is no unpaired electrons in the MO configuration of these species. So these are diamagnetic. O-2 is paramagnetic due to the presence of one unpaired electron.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
