How many electrons does iodine have Bohr model?
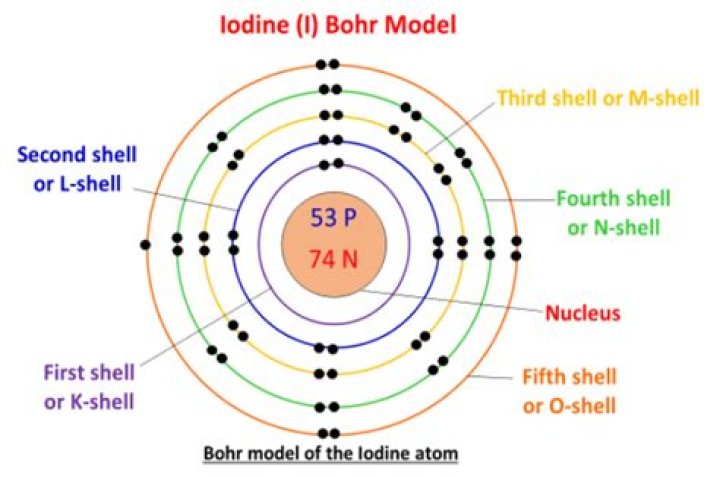
SolutionThe atomic number of iodine (53) tells us that a neutral iodine atom contains 53 protons in its nucleus and 53 electrons outside its nucleus. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 − 53 = 74).
Does iodine have 4 energy levels?
Explanation: Iodine, I , is located in period 5, group 17 of the periodic table, and has an atomic number equal to 53 . Notice that, in iodine’s case, the highest energy level is n=5 . You have two subshells occupied with electrons on this energy level, 5s2 and 5p5 .
What is the Bohr model for bromine?
The Bohr model of Bromine(Br) is drawn with four electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons, the third shell contains 18 electrons and the fourth shell contains 7 electrons.
How many neutrons does iodine 131 have?
A less stable form of iodine also has 53 protons (this is what makes it behave chemically as iodine) but four extra neutrons, for a total atomic weight of 131 (53 protons and 78 neutrons). With “too many” neutrons in its nucleus, it is unstable and radioactive, with a half-life of eight days.
What is the Bohr model of argon?
The Bohr model of Argon(Ar) is drawn with three electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons and the third shell contains 8 electrons. Argon is neutral and its atomic number is 18, hence, the number of protons and electrons available for its Bohr diagram is also 18.
What is the electronic configuration of 53?
Using our example, iodine, again, we see on the periodic table that its atomic number is 53 (meaning it contains 53 electrons in its neutral state). Its complete electron configuration is 1s22s22p63s23p64s23d104p65s24d105p5. If you count up all of these electrons, you will see that it adds up to 53 electrons.
How do you write iodine?
Iodine is an element with atomic symbol I, atomic number 53, and atomic weight of 126.90.
What is iodine on the periodic table?
iodine (I), chemical element, a member of the halogen elements, or Group 17 (Group VIIa) of the periodic table.
What element is in period 3 Group 17?
Chlorine is a halogen in group 17 and period 3. It is very reactive and is widely used for many purposes, such as as a disinfectant.
What is the Bohr model of chlorine?
The Bohr model of Chlorine(Cl) is drawn with three electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons and the third shell contains 7 electrons. Chlorine is neutral and its atomic number is 17, hence, the number of protons and electrons available for its Bohr diagram is also 17.
What does a Bohr model represent?
The Bohr model shows the atom as a central nucleus containing protons and neutrons with the electrons in circular orbitals at specific distances from the nucleus (Figure 1). These orbits form electron shells or energy levels, which are a way of visualizing the number of electrons in the various shells.
How is iodine-131 formed?
Iodine-131 is an artificially produced fission by-product resulting from nuclear weapons, above-ground nuclear testing, and nuclear reactor operations. Iodine-131 is found in the gaseous and liquid waste streams of nuclear power plants, but is not released to the environment during normal reactor operations.
What is the use of I-131?
I-131 is used in medicine to diagnose and treat cancers of the thyroid gland. Where does it come from? I-131 is produced commercially for medical and industrial uses through nuclear fission. It also is a byproduct of nuclear fission processes in nuclear reactors and weapons testing.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
