How many electrons does N2 have?
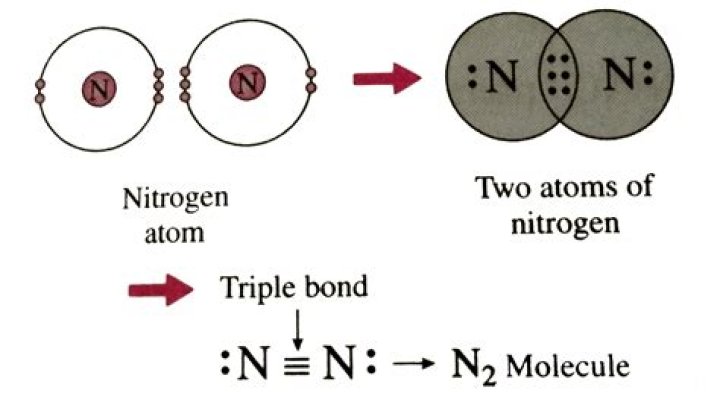
The electronic configuration of N2 is (σ1s) 2 (σ∗1s) 2 (σ2s) 2 (σ∗2s) 2 (π2px) 2 (π2py) 2 (σ2pz) the total number of electrons present in the nitrogen molecule (N 2 ) is 14. These 14 electrons can be accommodated in the various molecular orbitals in order of increasing energy.
How many electrons does N2 have?
That means there are 7 electrons in a nitrogen atom. Looking at the picture, you can see there are two electrons in shell one and five in shell two. ► More about the history and places to find nitrogen.
What is bond order of N2 and N2?
2, 2.5 and 3 respectively.
What is the shape of N2?
The molecular geometry of N2 is linear. N2 is colorless, odorless, and tasteless gas. Each nitrogen atom is surrounded by a lone pair of electrons.
What is the N2+ bond order when N2 is formed?
Bond order in N2 = 1/2 8 – 2 = 3 and bond order in N2+ =1/2 7-2 = 2.5Thus conversion of N2 to N2+ decreases bond order from 3 to 2.5 and hence increases the N-N bond distance.
How do you find the bond order of N2?
The Bond Order Formula can be defined as half of the difference between the number of electrons in bonding orbitals and antibonding orbitals. a = Number of electrons in bonding molecular orbitals. b = Number of electrons in antibonding molecular orbitals.
Is N2 linear or nonlinear?
Therefore, the molecular geometry for this molecule will be linear, with a bond angle of about 180∘ .
Is N2 a linear?
Nitrogen is a linear molecule. Every molecule that is made of two atoms has linear shape.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
