How many orbitals are half filled?
The p orbitals are half-filled; there are three electrons and three p orbitals. This is because the three electrons in the 2p subshell will fill all the empty orbitals first before pairing with electrons in them.
How many half filled orbitals are in a boron atom?
When boron is in a molecule with three regions of electron density, three of the orbitals hybridize and create a set of three sp2 orbitals and one unhybridized 2p orbital. The three half-filled hybrid orbitals each overlap with an orbital from a hydrogen atom to form three 蟽 bonds in BH3.
Which elements have half filled orbitals?
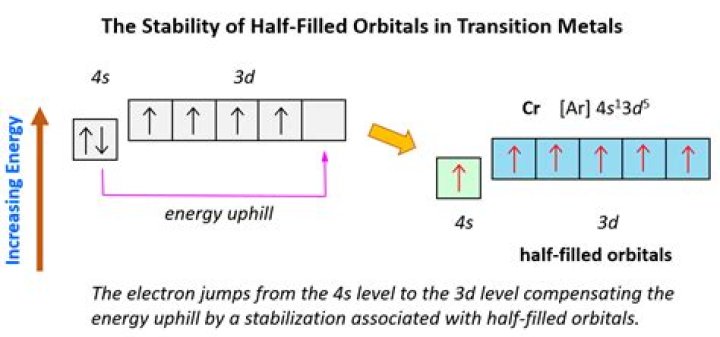
In the case of chromium, an electron from the 4s orbital moves into a 3d orbital, allowing each of the five 3d orbitals to have one electron, making a half-filled set of orbitals.
Does bromine have d orbitals?
Re: neutral bromine atom the d orbitals are completely filled. The electron configuration for Br is [Ar] 4s虏3d鹿鈦4p鈦. The d orbital is filled before the the 4p orbital because 3d orbital has less energy than 4p and the electrons fill up in order based on the orbital’s energy level.
What is half filled and fully filled orbitals?
– The reasons behind the greater stability of the atoms having half-filled or fully filled orbitals are symmetry and exchange energy. – We know that half-filled or fully filled atomic orbitals have more symmetry than any other electronic configuration and this symmetry leads to the greater stability of the atom.
How many half filled orbitals are in a Chromium atom?
So, now you should see that there are six unpaired electrons, meaning that you have six half-filled orbitals.
What is meant by partially filled orbitals?
Half filled orbitals and partially filled both are same terms and both are used for those orbitals having half number of electrons (orbital have one electron).
What is the number of electron of bromine?
The neutral atom of bromine has 35 electrons because the number of electrons equals the number of protons.
How many orbitals are filled in boron?
The next element after beryllium is boron. Since the 2s orbital is completely filled, a new type of orbital must be used for the fifth electron. There are three 2p orbitals available, and any of them might be used.
How many electrons does bromine need to fill s and p subshells?
To understand this principle, let’s consider the bromine atom. Bromine (Z=35), which has 35 electrons, can be found in Period 4, Group VII of the periodic table. Since bromine has 7 valence electrons, the 4s orbital will be completely filled with 2 electrons, and the remaining five electrons will occupy the 4p orbital.
Which atom does not contain partially filled p orbital?
Since it is part of the first period, helium does not have a p sublevel. Its filled 1s sublevel makes it very similar to the other members of Group 18.
Which is more stable half filled or partially filled?
The exactly half filled and completely filled orbitals have greater stability than other partially filled configurations in degenerate orbitals.
Which elements have a half filled p subshell?
All the elements belong to group 15 have half-filled p subshell. Thus, N, P, As, Sb, Bi and Mc all these elements belongs to 15 group, has
Which is the electron configuration for bromine quizlet?
the noble gas notation for the electron configuration of bromine is [Ar]3d10 4s2 4p5.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
