How many plane of symmetry are present in chcl3?
Trichloromethane belongs to the C3v Point group and contains;one C3 rotation axis along with 3蟽v planes of symmetry.
How do you find the symmetry number of a molecule?
molecule and that passes through the central atom. Therefore, The rotational symmetry number is given by the number of. permutations of m atoms of the same type divided by the.number of unique configurations, nd, that remain different. under rotation of the molecule, i.e.,蟽rot = m!nd. (5)
What is the symmetry number of ch4?
Symmetry: Td Symmetry Number 蟽 = 12.
Is the molecule CH3Cl chiral?
Chloromethane is not chiral, it is said to be achiral.
What does symmetrical mean in chemistry?
A symmetrical molecule is one whose appearance does not change if you turn it about an axis of symmetry; original and rotated states are indistinguishable from one another. Symmetry and asymmetry are important properties that affect how molecules respond to light waves, form bonds and operate biologically.
Does Bromochloromethane have a plane of symmetry?
Bromochloromethane has a plane of symmetry, and therefore it can be superimposed on its mirror image. It is achiral.
What is the symmetry number of h2?
In the context of atoms being perceived as point particles, this leaves hydrogen with a single E operation, thus symmetry number of one.
What is symmetry no?
The symmetry number or symmetry order of an object is the number of different but indistinguishable (or equivalent) arrangements (or views) of the object, i.e. the order of its symmetry group. The object can be a molecule, crystal lattice, lattice, tiling, or in a general case any mathematical object in N-dimensions.
What is the symmetry number of CO?
Statistical Thermodynamics
The symmetry number is the number of indistinguishable rotated positions. For a homonuclear diatomic such as H2, 蟽 = 2 since H鈥擧鈥 and H鈥测擧 are indistinguishable. For a heteronuclear diatomic, such as CO, 蟽 = 1 since C鈥擮 and O鈥擟 are distinguishable.
What is the point group of dichloromethane?
The answer is 3)has one C2 axis of rotation.
Dichloromethane whose structure is given below belongs to the C2v C 2 v point group.
What is the point group of ccl4?
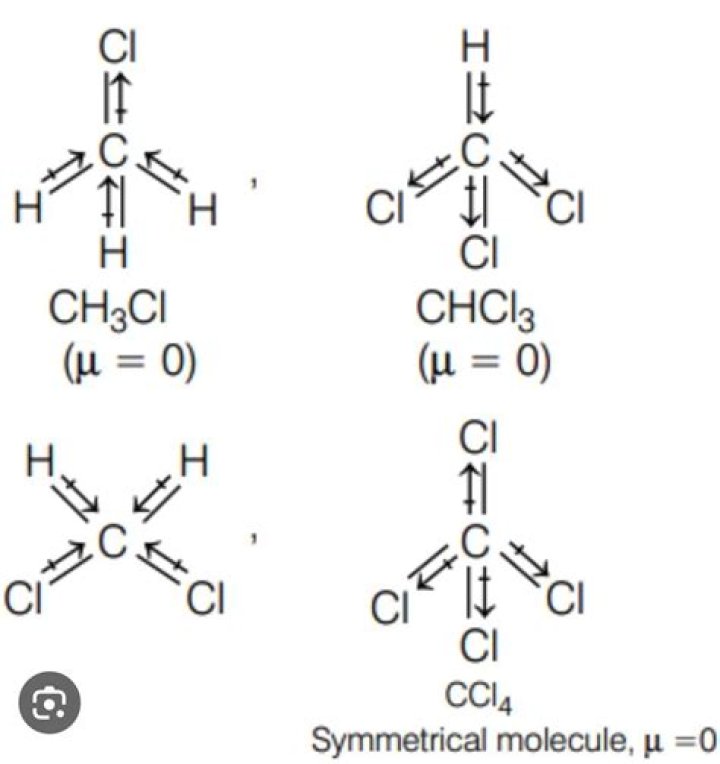
The molecule carbon tetrachloride belongs to the Td point group.
Is tetrahedral symmetrical?
Because a tetrahedron is a symmetric structure, any tetrahedral molecule where the corners of the tetrahedron are the same will be nonpolar. To find a polar molecule, just make one of the corners have a different polarity so that the dipoles do not cancel out!
What is the molecular geometry of CH3Cl?
Thus, CH3Cl’s molecular geometry is tetrahedral, and its bond angles are 109.5掳.
What do you mean by symmetry elements?
A symmetry element is a line, a plane or a point in or through an object, about which a rotation or reflection leaves the object in an orientation indistinguishable from the original.
Is CHCl3 polar or nonpolar?
CHCl3 is a polar molecule. Because of its tetrahedral molecular geometry shape and the difference in electronegativity value of carbon(C), hydrogen(H), and chlorine(Cl), CHCl3 is polar molecule.
What are the 4 types of symmetry?
Types of symmetries are rotational symmetry, reflection symmetry, translation symmetry, and glide reflection symmetry. These four types of symmetries are examples of different types of symmetry on a flat surface called planar symmetry.
What is c1 symmetry?
C1 Point Group
The point group C1 is the point group with the lowest symmetry. Molecules that belong to this point group only have the identity as symmetry element. It has no symmetry element, but the identity. The name C1 comes from the symmetry element C1. A C1 operation is the same as the identity.
How many symmetry elements are there?
There are 3 types of symmetry elements. They are: (a) centre of symmetry, (b) axis of symmetry and (c) plane of symmetry. A cube has 1 centre of symmetry. A cube has 3 axis of four fold symmetry, 4 axis of three fold symmetry and 6 axis of two fold symmetry.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
