How many valence electrons are present in the dot notation for radon?
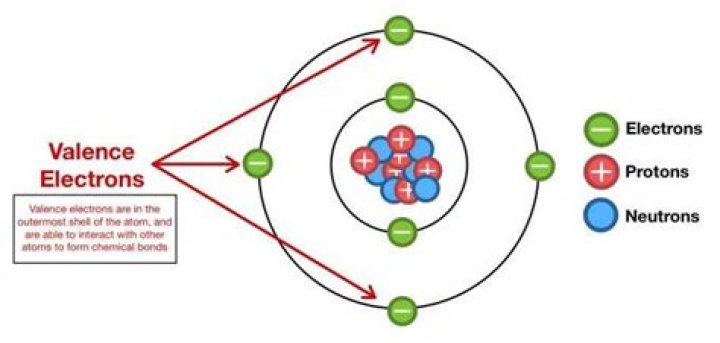
Radon has eight valence electrons.
How do you write a Lewis dot structure?
How to Draw a Lewis Structure
Step 1: Find the Total Number of Valence Electrons. Step 2: Find the Number of Electrons Needed to Make the Atoms “Happy” Step 3: Determine the Number of Bonds in the Molecule. Step 4: Choose a Central Atom. Step 5: Draw a Skeletal Structure. Step 6: Place Electrons Around Outside Atoms.
How many valence electrons should radium have in its Lewis dot model?
A radium atom has two valence electrons.
What is radon on the periodic table?
radon (Rn), chemical element, a heavy radioactive gas of Group 18 (noble gases) of the periodic table, generated by the radioactive decay of radium. (Radon was originally called radium emanation.) Radon is a colourless gas, 7.5 times heavier than air and more than 100 times heavier than hydrogen.
What is radon used for?
Radon decays into radioactive polonium and alpha particles. This emitted radiation made radon useful in cancer therapy. Radon was used in some hospitals to treat tumours by sealing the gas in minute tubes, and implanting these into the tumour, treating the disease in situ.
How many protons does radon have?
The chemical symbol for radon is Rn, and the mass number is usually placed either after the symbol (Rn-222) or to the left and above it (222Rn). In either case, it simply designates the element radon, which always has 86 protons, and that the particular isotope of radon in question is the one with 136 neutrons.
How do you draw a Lewis dot structure for a single element?
How to Draw a Lewis Dot Structure
Determine the total number of valence electrons to be depicted in the Lewis diagram. Place least electronegative element in center and draw single bonds from the central atom to other atoms.Determine how many electrons must be added to central element.
How many electron shells are in radium?
Radium Atomic and Orbital Properties
Radium atoms have 88 electrons and the electronic shell structure is [2, 8, 18, 32, 18, 8, 2] with Atomic Term Symbol (Quantum Numbers) 1S0.
What type of radiation is radium?
Most are due to gamma radiation, which can travel a long way through the air. Just being near high levels of radium is dangerous. Radium is a known cancer-causing substance. Exposure to high levels of radium can lead to higher chances of bone, liver and breast cancer.
How many core electrons are in radium?
Radium atoms have 88 electrons and the shell structure is 2.8. 18.32.
How does radon form?
Radon comes from the breakdown of naturally-occurring radioactive elements (such as uranium and thorium) in soils and rocks. The gas moves up through the soil to the surface, where it can enter homes, schools and the workplace through cracks and other holes in the foundation.
What does radon look like?
Radon (chemical symbol Rn) is an odorless, colorless, radioactive gas. It comes from the natural decay of uranium and radium found in nearly all rocks and soils. Radon can move up from the ground into buildings through openings in floors or walls that are in contact with the ground.
Why is it called radon?
Radon was discovered by Friedrich Ernst Dorn, a German chemist, in 1900 while studying radium’s decay chain. Originally named niton after the Latin word for shining, “nitens,” it has been known as radon since 1923.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
