how to calculate resonance energy, check these out | How do you calculate resonance energy of benzene?
The resonance energy is equal to the difference between the expected enthalpy and the calculated enthalpy. Complete step-by-step answer:The given value of the enthalpy of the benzene, i.e., C6H6 is -358.5 KJ /mol. This value can be written as expected enthalpy (ΔHexp).
How do you calculate resonance energy of benzene?
They give 977 kcal for the heat of atomization of the Kekul6 reference structure: Qaoo (Kekul6) = 3 X 56+ 3 X 95.2+ 6 X 87.3 = 977 kcal. The resonance energy of benzene based on this reference structure is therefore: QaoO(ac- tual) -QaoO(KekuI6) = 1039-977 = 62 kcal and not 39 kcal.
What is resonance energy explain with an example?
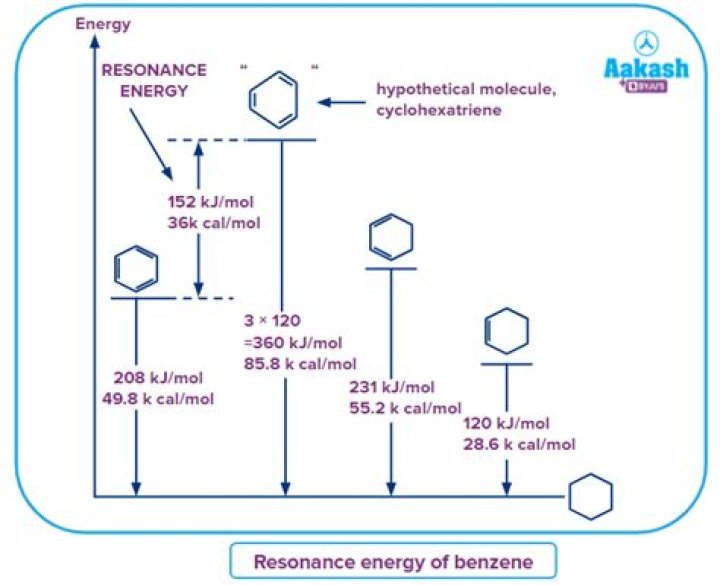
In molecules like benzene, resonance is also a quantum-mechanical kinetic energy effect. For example, the resonance energy of the six π-electrons in benzene is approximately 151 kJ/mol, less than half of the value we have calculated (per electron) in sodium metal.
What is resonance energy in organic chemistry?
Resonance energy: The theoretical difference in molecular energy between a resonance hybrid and the ‘most stable’ resonance contributor (if this resonance contributor existed as a real molecule). In other words, the stability gain by electron delocalization due to resonance versus the absence of such delocalization.
What is resonance energy in thermodynamics?
Resonance energy is the amount of energy required to convert the delocalized structure into a stable contributing structure. Delocalization occurs when the electric charge is spread over more than one atom.
What is resonance energy class 11?
What is resonance energy? It is defined as the difference in energy between the energy of the most stable of the contributing structures and energy of the actual molecule (resonance hybrid). Resonance Energy = Energy of most stable contributing structure – Energy of resonance hybrid.
How is resonance energy calculated by thermochemical data?
The topological resonance energy (TRE) of a catacondensed benzenoid hydrocarbon with h six-membered rings and K Kekulé structures can be calculated by the (approximate) formula TRE = Ah + B + CK e–Dh, where A = 0. 136, B = –0. 223, C = 0.
What are the rules of resonance?
Rules to remember for recognising resonance structures:
Atoms never move. You can only move electrons in π bonds or lone pairs (that are in p orbitals) The overall charge of the system must remain the same. The bonding framework of a molecule must remain intact.
What is resonance energy of naphthalene?
The resonance energy of Naphthalene is 30.5 Kcal/mol.
How do you calculate the resonance energy of n2o?
g —> N=N=O. Calculated value of ΔfH0 for this reaction will be ΔfH0 = [B.E. NΞN + ½ B.E.O=O] – [B.E.N=N + B.E.N=O] =[946 + ½ 498] – [418 + 607] kj mol-1 = 170kj mol-1 Resonance energy = Observed ΔfH0 – Calculated ΔfH0 = 82 – 170 = – 88 kj mol-1 .
What is meant by delocalization and resonance energy?
(i) Delocalisation: Delocalisation means that pairs of 71 electrons extend over 3 or more atoms. They belong to the whole molecule. For example, 6n electrons present in benzene are delocalised and are spread on the whole of the ring and this imparts extra stability to the molecule.
How do you calculate resonance energy of Class 11?
The resonance energy is equal to the difference between the expected enthalpy and the calculated enthalpy. Complete step-by-step answer:The given value of the enthalpy of the benzene, i.e., ${{C}_{6}}{{H}_{6}}$ is -358.5 KJ /mol. This value can be written as expected enthalpy ($Delta {{H}_{exp }}$).
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
