Is acetone a polar molecule?
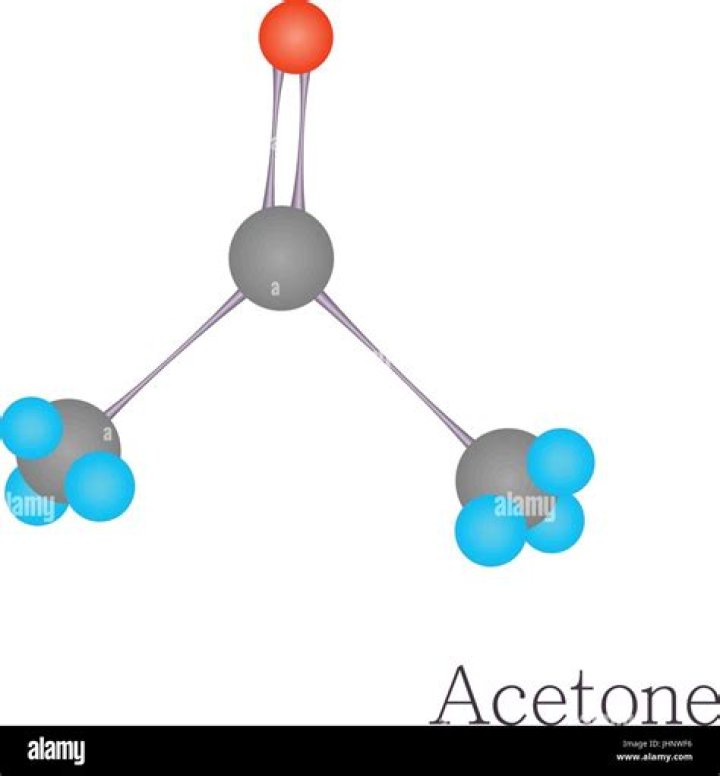
Acetone is a polar molecule because it has a polar bond, and the molecular structure does not cause the dipole to be canceled. … There is no other dipole to cancel out the C-O dipole. Conclusion: The molecule is polar.
What makes acetone polar?
The carbonyl group in acetone is polar, which is due to the difference in electronegativity (a chemical property that describes how strongly an atom will pull electrons toward itself) between the carbon and oxygen atoms. Because of this polarity, acetone enjoys mingling with polar solvents like water.
What type of molecule is acetone?
Acetone falls under the classification of ketones, which are organic compounds containing a carbonyl group bonded to two hydrocarbon groups. Acetone has a carbonyl group, where carbon and oxygen are bonded to each other with a double bond and is known as the simplest ketone.
Examples of Polar Molecules
Water (H2O) is a polar molecule. Ethanol is polar because the oxygen atoms attract electrons because of their higher electronegativity than other atoms in the molecule. Ammonia (NH3) is polar.Sulfur dioxide (SO2) is polar.Hydrogen sulfide (H2S) is polar.
How do you tell if a molecule is polar?
If the arrangement is symmetrical and the arrows are of equal length, the molecule is nonpolar.If the arrows are of different lengths, and if they do not balance each other, the molecule is polar.If the arrangement is asymmetrical, the molecule is polar.
Yes, we can say that NH3 is a polar covalent bond. Nitrogen forms a covalent bond with three atoms to form a molecule. You can get the difference between the N-H bond and the NH3 compound since the two are polar, even in their gaseous state.
What are examples of polar and nonpolar molecules?
Solution
Water is polar. Any molecule with lone pairs of electrons around the central atom is polar.Methanol is polar. Hydrogen cyanide is polar. Oxygen is nonpolar. Propane is nonpolar, because it is symmetric, with H atoms bonded to every side around the central atoms and no unshared pairs of electrons.
What causes a molecule to be polar?
Polarity results from the uneven partial charge distribution between various atoms in a compound. Atoms, such as nitrogen, oxygen, and halogens, that are more electronegative have a tendency to have partial negative charges. A polar molecule results when a molecule contains polar bonds in an unsymmetrical arrangement.
Acetone is a small molecule that has very non-polar and polar properties simultaneously. Its polar C=O. bond makes it miscible (soluble) in water, while its non-polar methyl (CH3) groups can interact with non-polar compounds.
Is acetone solution polar or nonpolar?
So, Is Acetone Polar or Nonpolar? Acetone is a polar substance because of polarity in the carbonyl group due to the difference in the electronegativity of oxygen and carbon atom. As a result, the dipole moment of Acetone is around 2.69 D.
Is acetone polar protic or polar aprotic?
Acetone is a polar aprotic solvent. A solvent is polar if it has a dipole moment greater than 1.6 D and a dielectric constant greater than 5. The values for acetone are µ = 2.88 D and ε = 21. So acetone is a polar solvent.
Is acetone heterogeneous or homogeneous?
Explanation: Acetone and water form a homogeneous mixture and not heterogeneous. They are miscible in each other in all proportions and so is air and water.
Acetone is a molecule with the molecular formula of C3H6O C 3 H 6 O . It is composed entirely of covalent bonds.
Is acetone chemically stable?
What are the stability and reactivity hazards of acetone? Chemical Stability: Normally stable. Conditions to Avoid: Open flames, sparks, static discharge, heat and other ignition sources.
Which molecule is most polar?
Water is the most polar molecule because a bond between oxygen and hydrogen has the most difference out of the atoms listed.
What is polar molecule give example?
A polar molecule has a net dipole as a result of the opposing charges (i.e. having partial positive and partial negative charges) from polar bonds arranged asymmetrically. Water (H2O) is an example of a polar molecule since it has a slight positive charge on one side and a slight negative charge on the other.
Lipids, i.e., fatty molecules, on the other hand, are non-polar, meaning that the charge distribution is evenly distributed, and the molecules do not have positive and negatively charged ends..
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
