Is acetone stronger than rubbing alcohol?
Acetone is the most powerful active ingredient in nail polish remover because it’s a more effective solvent than rubbing alcohol (especially ethanol).
Which is stronger acetone or rubbing alcohol?
Instead of being a form of alcohol, acetone is a ketone, and it is a much more effective solvent than rubbing alcohol. What this boils down to is that you can use pure acetone to remove nail polish from your nails, but it will be a lot more difficult to do so if you’re just using rubbing alcohol.
Can you use acetone as rubbing alcohol?
Honorable. Acetone will work fine. The whole point of using isopropyl is that it evaporates quickly, leaving no residue.
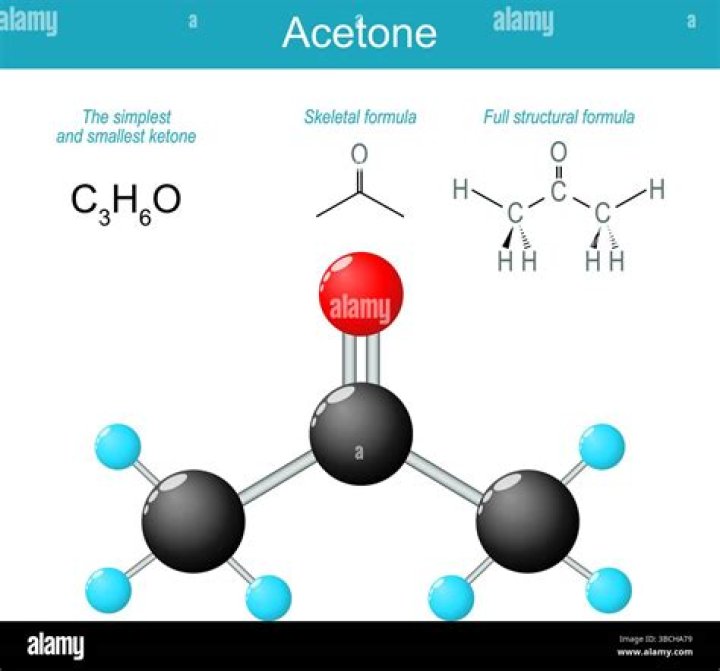
The key difference between acetone and isopropyl alcohol is that acetone has a C=O. bond in the middle of the chemical structure, whereas isopropyl alcohol has a C-OH group in the middle of the chemical structure.
What can I use in place of rubbing alcohol?
So what can you use as a substitute for rubbing alcohol? Soap and water, white vinegar and bleach are the best substitutes for rubbing alcohol for cleaning surfaces. For wound disinfection, something like hydrogen peroxide is the best alternative to rubbing alcohol.
It is prepared from a special denatured alcohol solution and contains approximately 70 percent by volume of pure, concentrated ethanol (ethyl alcohol) or isopropyl alcohol (isopropanol). Isopropyl alcohol is oxidized by the liver into acetone as a by product.
Does acetone sterilize?
“Acetone is a potent bactericidal agent and has considerable value for the routine disinfection of surfaces,” the study reported. To use nail polish remover to sanitize things in your home, be it beauty tools or other items, ideally, you would use 100 percent pure acetone nail polish remover.
Can I clean laptop with acetone?
Certain laptops, such as the white MacBooks, can get a nasty residue on places where your hands normally rest. Depending on the laptop, a little acetone (nail polish remover) can wipe away the mess. Start with just a dab to be on the safe side.
Acetone. One of the most popular solutions for cleaning electronics is acetone. Exposing circuit boards to this solvent for a prolonged time can potentially damage them, so you’ll want to ensure that you remove the acetone from your electronics as soon as possible during your cleaning routine.
Which evaporates faster ethanol or acetone?
While ethanol being an alcohol does have a direct O−H connection. Hence, ethanol has intermolecular hydrogen bonds. Therefore, more stroger physical bonds have to be destroyed in ethanol, than in acetone. Hence, acetone evaporates faster than ethanol inspite of having higher surface tension.
How does acetone react with alcohol?
Acetone reacts with alcohol, which is an organic compound with oxygen bonded to hydrogen, to form a hemiacetal. In this reaction, the oxygen on the alcohol goes after the carbon on the carbonyl group. Here, a ketone (like acetone) is exposed to a base, which removes hydrogen. The ketone then reacts with the halogen.
Can I use vodka instead of rubbing alcohol?
If you’re asking if you can use vodka in place of rubbing alcohol for cleaning, you’ll be pleased to find out that it’s possible. Both isopropyl alcohol and vodka are solvents that can mix with water. Their applications and properties are similar in many ways: Both isopropyl alcohol and vodka are good grease cutters.
Materials:
Water (distilled is recommended because you want your water to be free of any possible contaminants). 25 kilograms of Sugar per liter of water.1 packet of Yeast for every two liters of water.An Air Lock.
Is hydrogen peroxide the same as rubbing alcohol?
Rubbing alcohol can kill them within 10 seconds. Hydrogen peroxide is another antiseptic, or disinfectant, that kills viruses and various forms of bacteria. But it needs more time than rubbing alcohol does to kill germs. It needs up to 5 minutes to do its job.
Does acetone melt plastic?
If acetone comes into contact with the plastics below, you can expect a severe effect, especially at a high concentration and over a long time. The acetone will damage the plastic’s surface, softening it, smearing it, or even dissolving the plastic.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
