Is air a compound?
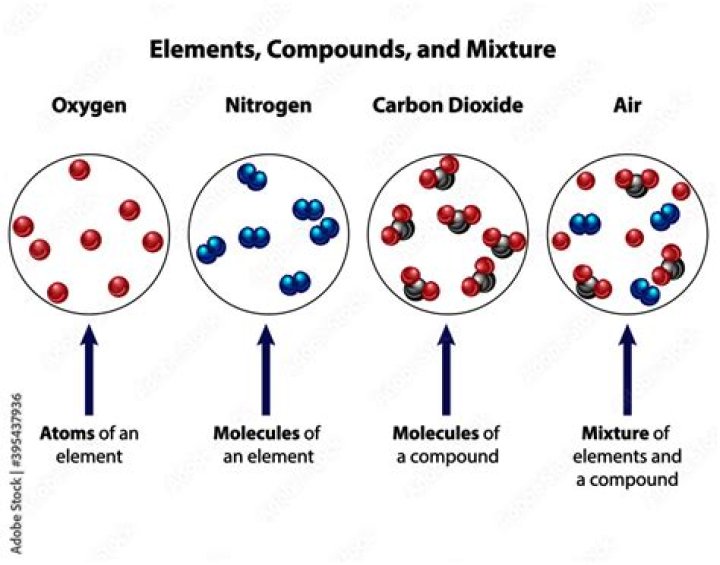
Air is a mixture but not compound. Its constituents can be separated. For example: oxygen, nitrogen etc. … Air shows properties same as the constituent gases present in it.
Is air homogeneous or compound?
Air is a homogeneous mixture of the gaseous substances nitrogen, oxygen, and smaller amounts of other substances. Salt, sugar, and substances dissolve in water to form homogeneous mixtures.
Is the air is a mixture?
Air is considered as mixture for the following reasons: Air can be separated into its components like nitrogen, oxygen, argon and carbon dioxide by the process of fractional distillation of liquid air. 2. Air shows the properties of all the gases present in the atmosphere.
Air is a solution made up of many gases. There is more nitrogen than any other gas in air, so it is considered the solvent in an air solution.
Why is air considered as a mixture and not a compound?
Air is a mixture and not a compound because: Air does not have a formula like a mixture, while compounds have a fixed formula. When air is formed by gases, there is no energy change. Components of air can be physically separated.
Air is mostly gas
The air in Earth’s atmosphere is made up of approximately 78 percent nitrogen and 21 percent oxygen. Air also has small amounts of lots of other gases, too, such as carbon dioxide, neon, and hydrogen.
Is there compound or mixture?
A compound contains atoms of different elements chemically combined together in a fixed ratio. A mixture is a combination of two or more substances where there is no chemical combination or reaction. Compounds contain different elements in a fixed ratio arranged in a defined manner through chemical bonds.
Why is air a heterogeneous mixture?
Heterogeneous mixture is always formed from two or more phases of matter. Also, they can be easily separated from each other. Hence, air is made up of various gases such as 78.09 Nitrogen, 20.95 oxygen, 0.93 argon,0.04 carbon dioxide and water vapors.
A substance that has a fixed chemical composition throughout is called a pure substance such as water, air, and nitrogen. A pure substance does not have to be of a single element or compound.
Why is air a mixture of gases?
Air is considered as a mixture because of the following reasons: > It is possible to separate air into its constituent gases by means of physical methods like fractional distillation. So, when liquefied air is distilled, nitrogen is separated out first and oxygen is left in the mixture.
Is air an example of heterogeneous mixture?
As long as each substance is mixed in enough to be indistinguishable from the others, it is a homogeneous mixture. A heterogeneous mixture is a mixture that does not have uniform composition throughout.
What kind of homogeneous is air?
Homogeneous mixture
In air, all gases would have a uniform composition. Therefore, the air is an example of homogeneous mixture.
The air around us is a mixture of gases, mainly nitrogen and oxygen, but containing much smaller amounts of water vapor, argon, and carbon dioxide, and very small amounts of other gases. Air is the commercial source for many of the gases it contains.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
