Is CF4 a polar or nonpolar molecule?
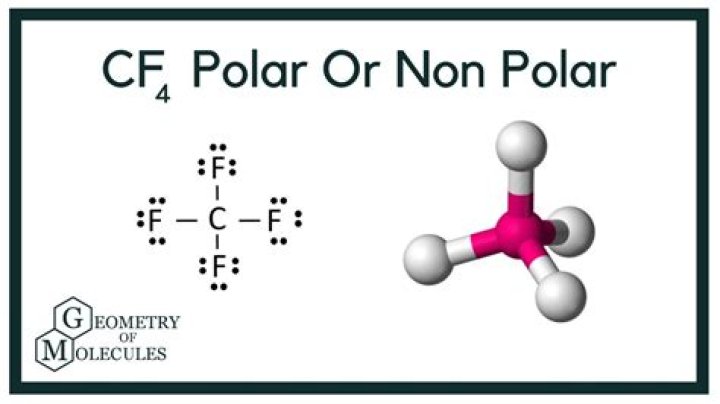
10.36 (a) CF4: Electron geometry-tetrahedral; molecular geometry-tetrahedral; bond angle = 109.5° Draw a Lewis structure for the molecule: has 32 valence electrons.
Is CF4 a polar or nonpolar molecule?
Carbon Tetrafluoride is a nonpolar covalent compound. If we look at the bonds individually, Carbon has an electronegativity of 2.5 and fluorine has an electronegativity of 4.0.
What is the molecular geometry for PF3?
PF3 has a trigonal pyramidal molecular geometry. Let’s count the areas around the phosphorus atom that
Why does CF4 have polar bonds?
Explanation: The carbon atom is sp3 hybridized, with the four C-F bonds pointing towards the corners of a tetrahedron. Each C-F bond is polar, because F is much more electronegative than C .
Why is CF4 a nonpolar molecule with polar bonds?
CF4 is in a tetrahedral molecular shape, so all the dipoles of the 4 polar C-F bonds cancel each other out, resulting in an overall nonpolar molecule. Think of it like a tug of war between four equally strong people, they don’t move anywhere as their forces cancel each other out!
How many bonding pairs are in CF4?
How many lone pairs of electrons appear in the Lewis structure of CF4? Each F atom has an octet consisting of three lone pairs and one bonding pair of electrons. There are 4 F atoms in CF4.
How many total bonds would be in a molecule of CF4?
Shared electron pairs are the electrons which participate in bonding. In CF4 C F 4 , there are total 8 shared electrons or 4 shared pair electrons which participate in bonding. Carbon makes 4 covalent bonds with fluorine atom.
Is CF4 a single bond?
A fluorine atom has seven valence electrons. Covalently sharing two electrons is also known as a “single bond.” Carbon will have to form four single bonds with four different fluorine atoms to fill its octet. The result is CF4 or carbon tetrafluoride.
How do you find the molecular geometry?
Steps Used to Find the Shape of the Molecule
Draw the Lewis Structure.Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons. Name the electron-group geometry. Looking at the positions of other atomic nuclei around the central determine the molecular geometry.
What molecular geometry would ph3 have?
In essence, ph3 is a Drago molecule and if we look at its bond angle data it shows that the p-orbitals have an angle of 90°. Looking at its Lewis structure we can state that molecular geometry of PH3 is trigonal pyramidal.
What is HCN molecular geometry?
Hydrogen cyanide has linear molecular geometry with bond angles of 180 degrees. As hydrogen and nitrogen tend to be far from each other, HCN forms a linear shape.
Why does CF4 have a dipole moment of 0?
The dipole moment arising from 3 C–F bonds present in the base of the tetrahedron cancels out the dipole moment arising from the 4th C–F bond. Hence the net or resulting dipole moment of CF4 is zero making the molecule non polar.
Why is CF4 nonpolar but SF4 is polar?
In VSEPR theory, the lone pair forces the molecular geometry of SF4 into a see-saw shape. Two of the S-F bonds are pointing away from each other, and their bond dipoles cancel. But the other two S-F dipoles are pointing “down”. Their bond dipoles do not cancel, so the molecule is polar.
Why do molecules follow a certain molecular geometry?
Molecules assume different shapes due to patterns of shared and unshared electrons. In these examples all electrons affecting the shape of the molecules are shared in the covalent bonds holding the atoms together to form the molecules.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
