Is CH2Cl2 a dipole?
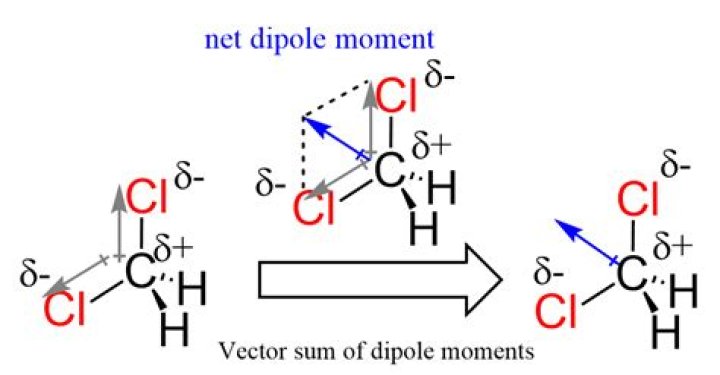
CH2Cl2 is a polar molecule due to its tetrahedral geometrical shape and difference between the electronegativity of Carbon, Hydrogen and Chlorine atoms. This develops a dipole moment across C-Cl and C-H bonds and the entire molecule results in a net 1.67 D dipole moment.
Does CH2Cl2 have a dipole moment?
The order of decreasing dipole moment is CH3Cl> CH2Cl2> CHCl3> CCl4. Hence, the dipole moment of CH2Cl2 is lower than the dipole moment of CH3Cl. Similarly, CHCl3 has a lower dipole moment than In CH2Cl2, The net dipole moment of CCl4 is zero because four C−Cl bond dipoles cancel each other.
Is CH2Cl2 nonpolar or polar?
Though a Chlorine atom is nonpolar, the polar molecule emerges after valence electrons of nonpolar molecules bond its properties. So, it is indeed a fact that even if there are nonpolar molecules, but the bonds do not cancel, and the geometry is showing polarity, then CH2CL2 is polar.
In CH2Cl2, the Cl pulls much more on the Carbon’s electrons (since it has higher electronegativity) than the Hydrogens, which sets up a dipole moment (i.e. polar).
What type of bond is CH2Cl2?
The bonds formed in Dichloromethane are covalent bonds. Central Carbon is hybridized as the molecule forms all the four bonds in the compound. An electron from the 22 orbital and three other electrons from 2p orbitals participate in forming bonds. Thus the hybridization of Carbon atom in CH2Cl2 is sp3.
Dichloromethane is a polar molecule as in it is a tetrahedral molecule, two and two bonds do not cancel each other. So, it shows a significant dipole moment. The interactions between the molecules are dipole-dipole interaction and London dispersion.
What is the direction of dipole moment in CH2Cl2?
Since it has a tetrahedral geometry and the dipole moment is a vector quantity, the vector sum of all dipole moments would try to cancel out. As they are in the outward direction, they will cancel to some extent. While in CH2Cl2, the C−H bond has the dipole towards C and there are only two C−Cl bonds.
Is CCl2H2 a dipole?
CCl2H2 is a bit confusing, but it most definitely has a dipole. It is a polar molecule.
CH2Cl2 polar molecule The molecule is tetrahedral due to 4 electron repulsions around the central C atom. Although the bond arrangement around the C atom in CH2Cl2 is symmetrical, the differing polarities of the C–H and C–Cl bonds means the effect of the polar bonds is not cancelled, so the molecule is polar.
Why is dichloromethane a polar molecule?
Dichloromethane is polar because it has different polarity bonds and its shape cannot arrange those bond dipoles to cancel out. Note that polarity can be considered for the whole molecule, functional group or particular bonds. E.g. CO2 has zero permanent dipole moment, as bond dipoles cancel each other.
Is dichloromethane a polar solvent?
Dichloromethane (DCM or methylene chloride) is an organochloride compound with the formula CH2Cl2. This colorless, volatile liquid with a chloroform-like, sweet odour is widely used as a solvent. Although it is not miscible with water, it is polar, and miscible with many organic solvents.
What is the bond angle of CH2Cl2?
So for example methane, CH4 and dichloromethane, CH2Cl2, will have the same tetrahedral geometry and 109.5º bond angles since they both have four bonding pairs and no non-bonding pairs of electrons around the central carbon.
Re: Polarity of CCl2H2
Since H and Cl have different electronegativities, the C-H and C-Cl dipole moments are also different. So although the 4 dipole moments do point in the same direction, they are not equivalent and thus do not cancel, making the overall molecule polar.
What type of solvent is dichloromethane?
Dichloromethane (DCM or methylene chloride) is an organochloride compound with the formula CH2Cl2. This colorless, volatile liquid with a chloroform-like, sweet odour is widely used as a solvent. Although it is not miscible with water, it is polar, and miscible with many organic solvents.
Does dichloromethane have dipole-dipole forces?
Dichloromethane can form dipole-dipole interactions between partially negative chlorine atoms and partially positive carbon atoms.
What intermolecular forces are present in ccl2h2?
Its strongest intermolecular forces are London dispersion forces. CH2Cl2 has a tetrahedral shape. The two C-Cl bond dipoles have a resultant that bisects the Cl-C-Cl bond angle.
Explanation: Whereas dipole-dipole interaction operates for methylene chloride , and dispersion forces are the primary intermolecular force in carbon tetrachloride .
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
