Is CH2CL2 polar?
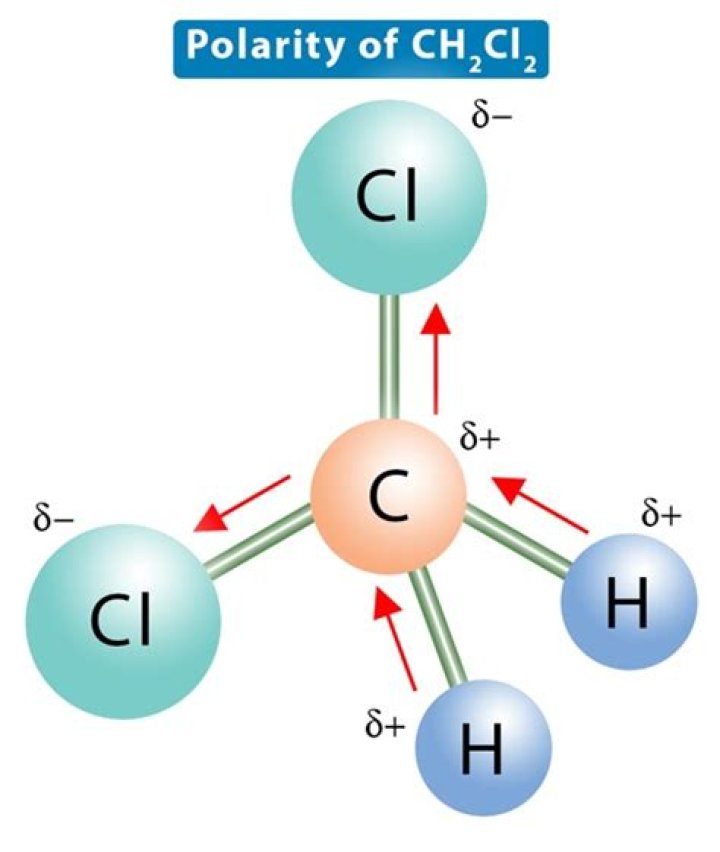
Though a Chlorine atom is nonpolar, the polar molecule emerges after valence electrons of nonpolar molecules bond its properties. So, it is indeed a fact that even if there are nonpolar molecules, but the bonds do not cancel, and the geometry is showing polarity, then CH2CL2 is polar.
Is C2h2cl2 polar?
C2h2cl2 hybridization Packaging. It has sp3 hybridization and polar. The bonds are considered to be nonpolar covalent (as all C-H bonds are incidentally) due to the low electronegativity difference between carbon (2.55) and hydrogen (2.20).
Is clch2ch2cl polar or nonpolar?
1,2-dichloroethane is a member of the class of chloroethanes substituted by two chloro groups at positions 1 and 2. It has a role as a non-polar solvent, a hepatotoxic agent and a mutagen.
Re: Why H2CBr2 polar? Answer: Tetrahedral molecules are nonpolar only when ALL the atoms bound to the central atom are the same atom type.
Why is dichloromethane a polar molecule?
Dichloromethane is polar because it has different polarity bonds and its shape cannot arrange those bond dipoles to cancel out. Note that polarity can be considered for the whole molecule, functional group or particular bonds. E.g. CO2 has zero permanent dipole moment, as bond dipoles cancel each other.
Dichloromethane (DCM or methylene chloride) is an organochloride compound with the formula CH2Cl2. This colorless, volatile liquid with a chloroform-like, sweet odour is widely used as a solvent. Although it is not miscible with water, it is polar, and miscible with many organic solvents.
How polar is dichloromethane?
So, Is CH2Cl2 polar or nonpolar? CH2Cl2 is a polar molecule due to its tetrahedral geometrical shape and difference between the electronegativity of Carbon, Hydrogen and Chlorine atoms.
Is ccl2h polar?
CCl2H2 is a polar molecule with a net dipole moment as shown on the right.
Question: For C2H2Cl2, if arranged with the two H next to each other and the two Cl next to each other (in cis arrangement), it results in a polar molecule.
Which of the two forms of C2H2Cl2 is polar?
trans-1,2-dichloroethene has no dipole moment and is nonpolar, while cis-1,2-dichloroethene has a dipole moment and is polar.
What intermolecular forces are present in C2H2Cl2?
London/Dispersion forces.Ion-dipole forces.Dipole-dipole forces.Hydrogen bonding.Metallic bonding.
Is dichloromethane more polar than water?
Yeh. CH2Cl2 is barely polar actually since C-Cl has deltaEN=0.5 where C-H gives a 0.4. Much less polar than water for sure..
CH2BR2 is a polar molecule because its dipole moments do not cancel out so it has a non-zero net moment.
Is CH2Cl2 asymmetric or symmetrical?
Although the bond arrangement around the C atom in CH2Cl2 is symmetrical, the differing polarities of the C–H and C–Cl bonds means the effect of the polar bonds is not cancelled, so the molecule is polar.
What shape is CH2Cl2?
The CH2Cl2 molecule is polar in nature. As the shape of the molecule is tetrahedral and Carbon and Chlorine have a difference in their electronegativity.
What is the bond angle of CH2Cl2?
So for example methane, CH4 and dichloromethane, CH2Cl2, will have the same tetrahedral geometry and 109.5º bond angles since they both have four bonding pairs and no non-bonding pairs of electrons around the central carbon.
PH3 is a polar molecule because it has a bent structure due to lone pairs of electrons and electron-electron repulsion. Phosphorus’s electronegativity is a nonpolar molecule because it is the same, but since Phosphorus has a lone pair, PH3 is a polar molecule.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
