Is CH3CH2OH polar or nonpolar or ionic?
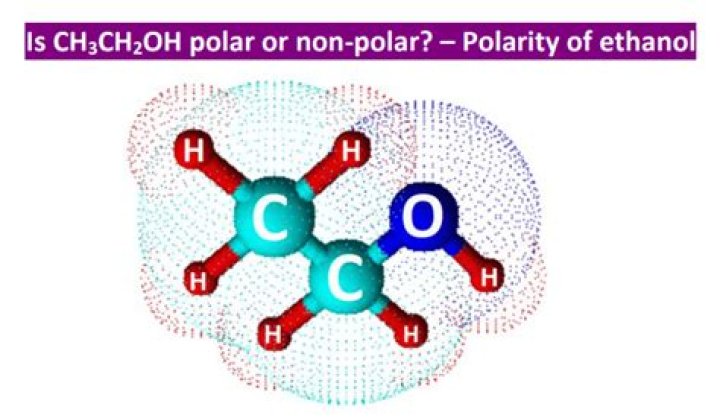
Thus it is polar.
Is CH3CH2OH polar or nonpolar or ionic?
The structure of ethanol is given below. Hence, the molecule CH3CH2OH C H 3 C H 2 O H is polar.
Is hc3 polar?
Ethane is non-polar, due to 2 reasons: There is no difference in electronegativity between the 2 carbons, and the difference in electronegativity between C and H is minimal .
Is dimethyl ether polar or non polar?
The C-O bonds of methoxymethane (dimethyl ether) (CH3-O-CH3) are polar. The geometry of the molecule is angular, resulting in an overall molecular dipole. Hence the molecule will be subject to dipole-dipole and dipole/induced dipole interactions as well as the stronger dispersion forces.
Is CH3OCH3 polar or nonpolar molecule?
Is CH3OCH3 a polar or a nonpolar molecule? – Quora. It is a polar molecule . Though it might seem that it is non polar because the dipole moment will be cancelled due to symmetry but actually it has no symmetry .
How polar is CH3CH2OH?
#NAME?
Is CHCl3 polar?
CHCl3 is a polar molecule. Because of its tetrahedral molecular geometry shape and the difference in electronegativity value of carbon(C), hydrogen(H), and chlorine(Cl), CHCl3 is polar molecule.
Why is dimethyl ether slightly polar?
Ethers are small dipoles due to the electronegativity difference between carbon and oxygen. As both dipole moment vectors point roughly in the same direction, they do not cancel each other out like in the case of CO2, so the molecule has a net dipole moment and is therefore (weakly) polar.
Which is more polar ethanol or dimethyl ether?
Because hydrogen bonds are typically much stronger attractions than ordinary dipole moments, a group of ethanol molecules is much harder to separate from each other than a group of dimethyl ether molecules. The ethanol has a much higher boiling point.
Is an ether polar?
The C – O bonds in ether are polar and thus ethers have a net dipole moment. The weak polarity of ethers do not appreciably affect their boiling points which are comparable to those of the alkenes of comparable molecular mass. Ethers have much lower boiling points as compared to isomeric alcohols.
Is dichlorodifluoromethane polar?
Information on this page: Normal alkane RI, non-polar column, custom temperature program.
Why is CH3COCH3 polar?
Differences in electronegativity help determine whether a compound is polar or no. The oxygen linked to the carbon has a tendency to attract the pair of bond electrons between the oxygen and carbon, so CH3COCH3 is polar.
Is CH3CH2CH3 soluble in water?
Insoluble: CH3CH2CH3 is nonpolar that cannot form hydrogen bonding attraction with O-H diploes in water which is more polar molecule.
What shape is CH3CH2OH?
In methanol, CH3CH2OH, there are three interior atoms: two C and one O. We assign the molecular (and electron-domain) geometry about each interior (central) atom separately. The geometry around the first C is tetrahedral. The geometry around the second C is tetrahedral.
What type of chemical bond is CH3CH2OH?
The hydrogen-oxygen and carbon-oxygen bonds are polar covalent bonds.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
