Is CH3Cl polar or nonpolar?
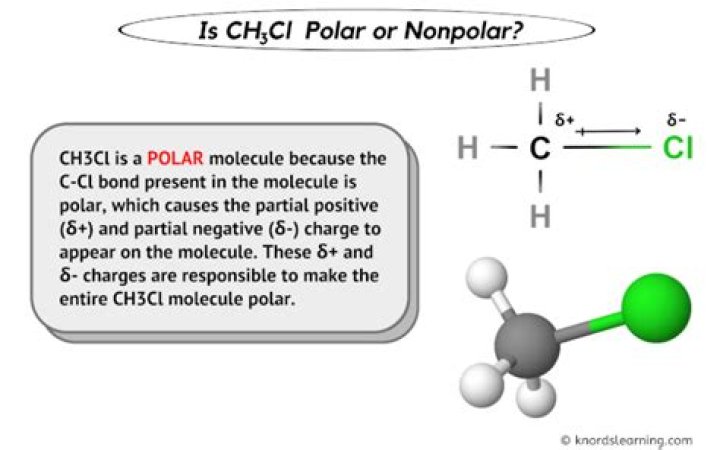
Because the C-Cl bond is polar, the CH3Cl has a net dipole, so CH3Cl is polar.
Is CH3Cl polar or nonpolar atom closest to negative side?
So, Is CH3Cl polar or non-polar? Yes, Methyl chloride (CH3Cl) or Chloromethane is a polar molecule. The C-Cl covalent bond shows unequal electronegativity because Cl is more electronegative than carbon causing a separation in charges that results in a net dipole.
Why is CH3Cl polar and CCl4 nonpolar?
The four bonds of carbon tetrachloride (CCl4) are polar, but the molecule is nonpolar because the bond polarity is canceled by the symmetric tetrahedral shape. When other atoms substitute for some of the Cl atoms, the symmetry is broken and the molecule becomes polar. In this case, chloroform is considered non-polar.
The order of decreasing dipole moment is CH3Cl> CH2Cl2> CHCl3> CCl4. Hence, the dipole moment of CH2Cl2 is lower than the dipole moment of CH3Cl. Similarly, CHCl3 has a lower dipole moment than In CH2Cl2, The net dipole moment of CCl4 is zero because four C−Cl bond dipoles cancel each other.
Is ch2cl3 polar or nonpolar?
Dichloromethane is polar because it has different polarity bonds and its shape cannot arrange those bond dipoles to cancel out. Note that polarity can be considered for the whole molecule, functional group or particular bonds.
The difference in electronegativity of the carbon and chlorine is 0.5. Therefore, the bond between carbon and chlorine is polar covalent. The structure for molecule is as follows: Since the three polar bonds are not symmetrical, they do not cancel the dipole each other.
Is CH3Cl ionic or covalent?
CH3Cl is covalent as no metals are involved. Both ionic and covalent bonds shared the fixed quantities during bond formation. 5) CH2Cl2 covalent bond.
Which of the following molecules are polar CH3Cl?
Methyl chloride is a polar molecule because of the electronegativity difference between carbon and chlorine atom.
Suppose you compare the electronegativities of both the atoms carbon and Chlorine. In that case, you will find out that the chlorine atom is more electronegative than the carbon atom as it is closer to Flouirne on the periodic table.
Is sibr4 polar or nonpolar?
The electronegativity of silicon is 1.8, that of bromine is 2.8. The silicon – bromine bond is polar. Showing these bonds as arrows in a tetrahedral structure clarifies that silicon tetrabromide is a nonpolar molecule.
Is CH3Cl a dipole dipole?
CH3Cl intermolecular forces has dipole-dipole forces and London dispersion forces. the chemical name of CH3Cl is chloromethane.
Is CH3Cl chiral or achiral?
Chloromethane is not chiral, it is said to be achiral. What if one of the hydrogen atoms in chloromethane is substituted for a different atom, an atom of bromine for example.
The (HCH) bond angle in CH3Cl is 110.5°. The HCH bond angle is wider than in CH4 which suggests that the C–Cl single bond takes up less space close to the carbon. This is due to the C–Cl bonds polarity with δ– on the chlorine, chlorine being more electronegative than carbon (and hydrogen).
How many covalent bonds are present in CH3Cl?
Therefore, all of these share electrons and as a result, carbon forms 3 covalent bonds with hydrogen and one covalent bond with chlorine.
How many bonding electrons does CH3Cl?
In the case of CH3Cl, the total number of valence electrons will be 14. Step 3: Figure out how many more electrons are required to stabilize one CH3Cl molecule: One CH3Cl molecule needs 8 more electrons to stabilize its structure completely.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
