Is CS2 a polar covalent bond?
CS2 (Carbon disulfide) is nonpolar because of its symmetric (linear) shape. Although carbon and sulfur differ in their electronegativity and C-S bond is polar, the polarity of both opposite C-S bonds gets canceled by each other resulting in a nonpolar molecule.
What type of covalent bond is CS2?
The CS2 molecule is a nonpolar covalent bond with two double bonds. The valence electrons that form this type of bond are in 2s and 2p orbitals of the carbon atom.
What is an example of a polar covalent bond?
Answer: Polar covalent bond is a type of chemical bond where one pair of electrons is shared unevenly between two atoms. For example, Hydrogen chloride (HCl) molecule. The bonding of hydrogen and chlorine atoms leans more towards Cl atoms because Cl is more electronegative in nature than hydrogen.
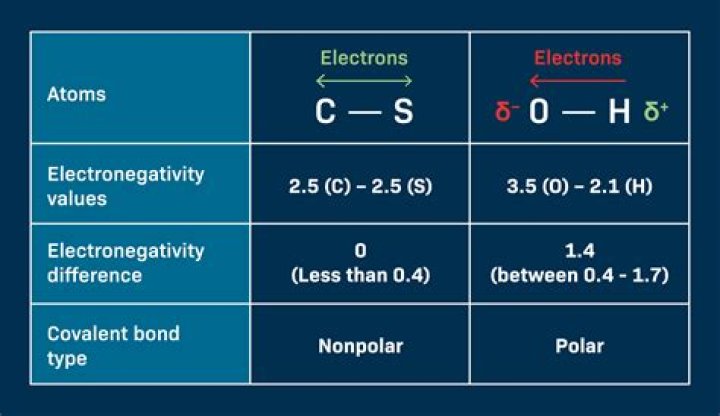
Although C and S have very similar electronegativity values, S is slightly more electronegative than C, and so the C-S bond is just slightly polar.
Is CS2 a polar molecular or a nonpolar molecule?
CS2 C S 2 is a nonpolar molecule is a nonpolar molecule . Explanation: CS2 C S 2 , similarly to CO2 C O 2 , is a linear molecule
So, is CS2 ionic or covalent? CS2 is a covalent molecule as both the carbon and sulfur atoms have nearly the same electronegativity values. The electronegativity difference among the carbon and sulfur atom is nearly 0.03, and thus the bond between them is hardly even polar.
How many polar bonds does CS2 have?
In CS2 molecule, two double bonds are formed consisting of eight valence electrons. Thus it takes up eight valence electrons out of 16 valence electrons. These valence electrons that form the double bond with the Carbon atom are in 2s and 2p orbital of the Carbon atom.
Is CS2 polar or nonpolar?
So, is CS2 polar or nonpolar? CS2 (Carbon disulfide) is nonpolar because of its symmetric (linear) shape. Although carbon and sulfur differ in their electronegativity and C-S bond is polar, the polarity of both opposite C-S bonds gets canceled by each other resulting in a nonpolar molecule.
If the electronegativity difference between the two atoms is between 0.5 and 2.0, the atoms form a polar covalent bond.
Examples of polar molecules include:
Water – H2O.Ammonia – NH. Sulfur dioxide – SO. Hydrogen sulfide – H2S.Ethanol – C2H6O.
How do you know if a bond is polar covalent?
The terms “polar” and “nonpolar” usually refer to covalent bonds. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; if the result is between 0.4 and 1.7, then, generally, the bond is polar covalent.
Is SIF polar covalent?
Despite the large difference in electronegativity between Si and F , SiF4 the electronegativity doesn’t matter because of the tetrahedral of four Si-F make the the compound nonpolar.
What intermolecular forces are present in CS2?
Sample Response: CS2 and COS both have London Dispersion Forces, but since COS is a polar molecule, it also exhibits dipole-dipole forces. However, the London Dispersion Forces in CS2 are so strong that they overpower the strength of both the LDFs and the dipole-dipole forces in COS.
Carbon disulfide is not a polar molecule. Electronegativity is the measure of how strongly an atom will attract electrons to itself.
What is the geometry and polarity of the carbon disulfide molecule?
The molecular geometry of CS2 is linear with symmetric electron region distribution around the central atom. Therefore this molecule is nonpolar.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
