Is freezing point a physical or chemical property?
For example, the freezing point of a substance is a physical property: when water freezes, it’s still water (H2O)—it’s just in a different physical state.
Is freezing a chemical or physical change?
Freezing and boiling are considered to be examples of chemical reactions. This depends on their conception of substance. If students regard ice as a different substance from liquid water they are likely to classify the melting of ice as a chemical change.
Is freezing chemical property?
Other physical properties, such as the melting temperature of iron or the freezing temperature of water, can only be observed as matter undergoes a physical change. The change of one type of matter into another type (or the inability to change) is a chemical property.
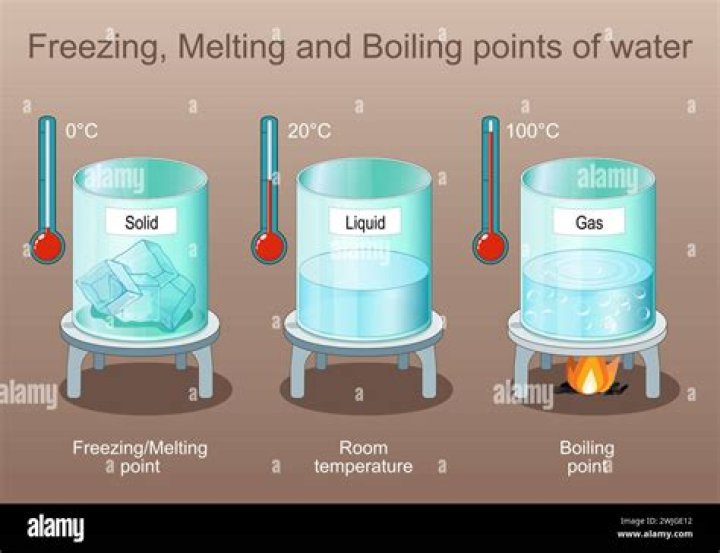
Physical properties are characteristics of a substance. They do not change. Physical properties include color, smell, freezing/melting point, and density.
Why is freezing a physical property?
Freezing, or solidification, is the withdrawal of heat from a substance to change that substance from a liquid to a solid. The temperature must be below the substance’s freezing point for the change to occur. Turning water into ice using a freezer is an example of this physical change.
Freezing and melting are physical changes. The substances produced during chemical changes however cannot easily change back into the original substances.
Is freezing of water chemical or physical?
Water boiling, melting ice, tearing paper, freezing water and crushing a can are all examples of physical changes. On the other hand, chemical changes are a bit different. In a chemical change, a new substance is formed. The chemical change also usually involves heat, burning, or other interaction with energy.
Is freezing point an intensive physical property?
Thus, freezing point is an intensive property and doesn’t change when we vary amount. Other examples of intensive properties are color, boiling point, pressure, molecular weight and density. Density is an interesting example.
Physical properties of matter include color, hardness, malleability, solubility, electrical conductivity, density, freezing points, melting points, and boiling points.
Is melting point a physical property or a chemical property?
Characteristics such as melting point, boiling point, density, solubility, color, odor, etc. are physical properties. Properties that describe how a substance changes identity to produce a new substance are chemical properties.
Why is melting point considered a physical property?
Physical changes are related to physical properties since some measurements require that changes be made. Melting Point: As solid matter is heated it eventually melts or changes into a liquid state at the melting point. Ice (a solid form of water) melts at 0 oC and changes to the liquid state.
What are the chemical properties of ice?
Because they share a common composition with their liquid state, ice molecules also consist of the same 2 to 1 ratio of hydrogen and oxygen atoms, the H2O molecule. The shape of this molecule, the oxygen atom at the center with the two hydrogen atoms separated by an angle of 104.52°, dictates the structure of ice.
Familiar examples of physical properties include density, color, hardness, melting and boiling points, and electrical conductivity. Other physical properties, such as the melting temperature of iron or the freezing temperature of water, can only be observed as matter undergoes a physical change.
What is the freezing point of a substance?
The freezing point of a substance is the temperature at which the solid and liquid forms are in equilibrium. To reattain equilibrium, the freezing point of the solute and solvent mixture is lowered relative to the original pure solvent.
What happens at the freezing point of a substance?
freezing point, temperature at which a liquid becomes a solid. As with the melting point, increased pressure usually raises the freezing point. Some liquids can be supercooled—i.e., cooled below the freezing point—without solid crystals forming.
Is a lake freezing a chemical change?
Matter can change form through physical and chemical changes, but through any of these changes matter is conserved. To change between these states, water must undergo physical changes. When water freezes, it becomes hard and less dense, but it is still chemically the same.
Even processes as simple as changes in phase (solid to liquid, liquid to gas, etc.) are really chemical in nature. In the process of freezing to ice, the molecules in liquid water start in one configuration, form bonds as they take on the new configuration, and release energy in the process.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
