Is HClO weak electrolyte?
Weak Acid: dissolves but less than 100% dissociates to produce protons (H+) 1. any acid that is not one of the seven strong is a weak acid (e.g. H3PO4, HNO2, H2SO3, HClO, HClO2, HF, H2S, HC2H3O2 etc.) 2.
Is HClO strong or weak?
These acids are the only ones that fully dissociate in water. All other acids are weak acids. Since HClO is not one of these seven, and there is no -OH group present as there is in bases, HClO is a weak acid.
Is HOCl a strong electrolyte?
Hypochlorous acid (HOCl or HClO) is a weak acid that forms when chlorine dissolves in water, and itself partially dissociates, forming hypochlorite, ClO−.
Why HClO is a weak acid?
HClO is an acid as is has the proton that it can donate but it is a weak acid because it is not one the acid amoung the list of the strong acids.
How weak is HClO?
Hypochlorous acid, HClO, is a weak acid with Ka = 3.0 x 10-8.
Why is HOCl an acid?
What is HOCl? HOCl is a weak acid that is formed when chlorine (a gas) dissolves in water. It is used as an active ingredient in sanitizers and disinfectants because of its ability to break apart cell membranes, similar to the mechanism of action of sodium hypochlorite (bleach) or hydrogen peroxide.
What type of acid is HClO?
Hypochlorous acid is a chlorine oxoacid with formula HOCl; a weak, unstable acid, it is the active form of chlorine in water. It has a role as a human metabolite, an EC 3.1. 1.7 (acetylcholinesterase) inhibitor and an EC 2.5.
Is HOCl and HClO the same?
HOCl and HClO are exactly same. This is molecular formula of hypochlorus acid. It is an oxyacid of chlorine molecule. It is a weak acid which gives .
Does HClO dissociate in water?
Hypochlorous acid (HClO) is formed when chlorine dissolves in water, and it quickly dissociates into hypochlorite (ClO.
Is CuSO4 a strong electrolyte?
Two compounds that are strong electrolytes are the ionic compounds ZnSO4 and CuSO4. If the compound is either one of these, then it is definitely a strong electrolyte.
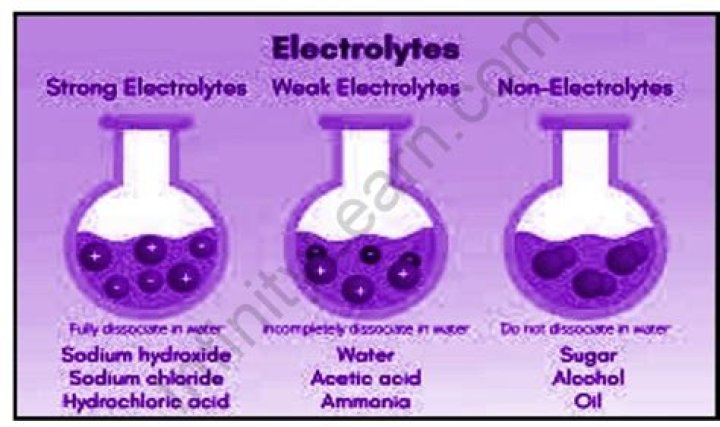
What substances are strong electrolytes?
Strong Electrolytes
hydrochloric acid, HCl.hydroiodic acid, HI.hydrobromic acid, HBr.nitric acid, HNO3sulfuric acid, H2SO4chloric acid, HClO3perchloric acid, HClO4
What are the non-electrolytes?
A non-electrolyte is a solute that does not dissociate, or separate, into ions during the dissolving process. Therefore, solutions that are generated through the solvation of non-electrolytes do not contain ions and, instead, are comprised exclusively of neutral molecules.
Is glycerol an electrolyte?
In this study, a GPE based on PEO and glycerol is investigated for the first time as electrolyte for environmentally friendly DSSCs with natural dyes. A glycerol electrolyte without PEO had higher ionic conductivity and achieved higher efficiencies as well but leaked from the unsealed DSSCs.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
