Is HCN a covalent bond?
HCN with ten valence electrons is a molecular bond with a linear-shaped molecule, also known as a covalent bond. … Since HCN contains N, which is not directly bonded to hydrogen atoms, it has dipole-dipole forces act between the electron pair of the HCN molecule.
Is HCN ionic or covalent?
Accordingly, carbon and hydrogen share electrons of each other and forms a covalent bond(C-H) whereas carbon and nitrogen form a triple bond (C≡N) to share their three electrons with each other. As a result, the molecule H-C≡N becomes stabilized.
What kind of bond is HCN?
HCN, hydrogen cyanide, is a volatile and poisnous compound with distinguished bitter odor. It is linear molecule with a triple bond between C and N atom and has bond angle of 180 degrees.
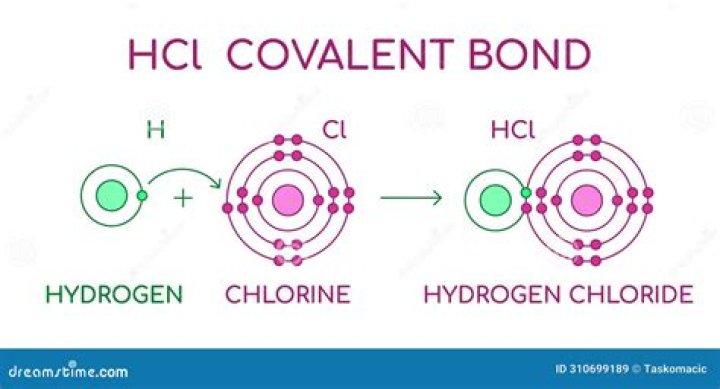
HCl is a covalent compound because the electronegativity difference between hydrogen and chloride is less than 2.0. The H-Cl bond will act as a dipole with a partial positive charge and a partial negative charge on the hydrogen atom and chlorine atom, respectively, in the HCl molecule.
What type of bond is covalent?
Forming Covalent Bonds
Covalent bonds are a class of chemical bonds where valence electrons are shared between two atoms, typically two nonmetals. The formation of a covalent bond allows the nonmetals to obey the octet rule and thus become more stable. For example: A fluorine atom has seven valence electrons.
Bond formation, hydrogen cyanide molecule. This is an example of covalent bonding, with the single bond (left) and the triple bond (right) formed by shared electron pairs, each pair consisting of an electron from each atom. The outer electron shell of each atom is shown as a blue ring.
Is HCN a dipole?
(d) HCN is a linear molecule; it does have a permanent dipole moment; it does contain N, however the nitrogen is not directly bonded to a hydrogen. Therefore dispersion forces and dipole-dipole forces act between pairs of HCN molecules.
Is HCN polar covalent or nonpolar covalent?
HCN, or hydrogen cyanide, is a polar molecule because there is a large electronegative difference between the N and H across the linear molecule. It consists of two polar bonds whose polarities line up in the same direction. The Lewis structure determines whether an entire molecule is polar or essentially nonpolar.
In HCN, there are two sigma bonds, C-H and C-N. The number of sigma bonds is equal to the number of hybrid orbitals formed. So the hybridization for HCN molecule is sp hybridization.
Is Cao a covalent compound?
Calcium oxide is ionic because it is formed between a metal and non-metal and bonds formed between a metal and non-metal atoms are ionic. Bonds formed between two molecules that are coordinate covalent bonds.
Is H2 a covalent bond?
Covalent molecules made of only one type of atom, like hydrogen gas (H2), are nonpolar because the hydrogen atoms share their electrons equally.
Is LIBR covalent?
Lithium bromide is an ionic compound of lithium and bromine.
Noncovalent bonds determine the shape of many large biological molecules and stabilize complexes composed of two or more different molecules. There are four main types of noncovalent bonds in biological systems: hydrogen bonds, ionic bonds, van der Waals interactions, and hydrophobic bonds.
What are the 4 types of covalent bonds?
Types of Covalent Bonds
Single Covalent Bond.Double Covalent Bond.Triple Covalent Bond.
What is a covalent bond Class 10?
A covalent bond is a chemical bond in which pairs of electrons are shared between two atoms. The covalent bond is also called a molecular bond.
Is HCN an electrolyte?
HCN is classified as a weak electrolyte.
This is because when dissolved in water, a low percentage of the HCN molecules ionize. HCN is not one of the strong acids and thus would be one of the weak acids (which are weak electrolytes).
The molecule hydrogen cyanide, HCN, does not have hydrogen bonding.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
