Is NF3 sp3 hybridization?
NF3 is predominantly covalent in nature and has pyramidal structure ( the central atom is sp3 hybridised) with a lone pair of electrons in the fourth orbital.
Is NF3 sp3 hybridization?
Hence, in the $N{F_3}$ molecule there are three bond pairs and one lone pair. Nitrogen atoms are $N{F_3}$ molecules and are supposed to be [s{p^3}] hybridization. As we discussed above, the hybridisation of nitrogen atoms in $N{F_3}$ molecules. So, the correct option is $left( A right)$ .
What is the shape and hybridization of NF3?
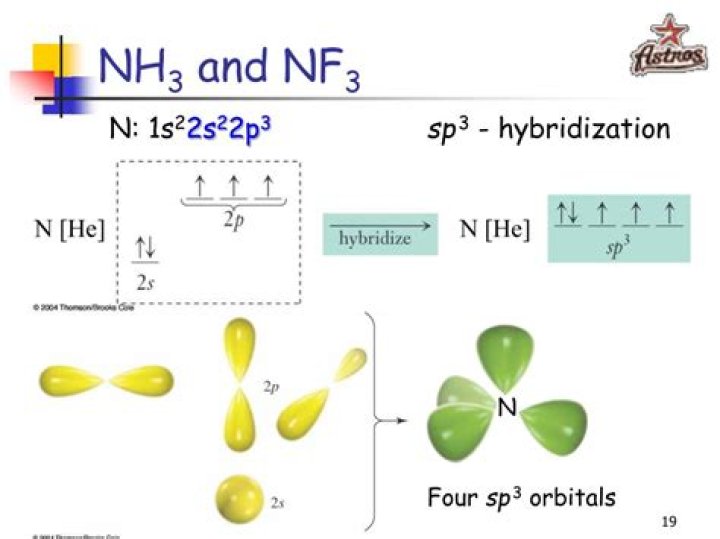
Hybridization of NF3 is Sp³. NF3 is polar in nature. The molecular geometry or shape of NF3 is a trigonal pyramid and its electron geometry is tetrahedral. NF3 lewis dot structure contains 1 lone pair and 3 bonded pairs.
How do you find the hybridization of NF3?
The electronic configuration of central atom nitrogen is 2,5.
→ Three flourine atoms will overlap with three sp3 hybrid orbitals.→ While, in one sp3 hybrid oribital, a lone pair will be present.So, hybridization of central atom in NF3 is sp3.
What is the geometry of NF3?
The F-N-F bond angle is 97 degrees in the trigonal pyramidal NF3 molecular geometry. The NF3 molecule has a trigonal pyramidal geometry shape because it contains three fluorine atoms in the geometry and four corners with one lone pair of electrons.
What is dipole moment NF3?
Answer: N- atom is the central atom of NF3 and NH3. As, F-atom is more electronegativity than H- atom,NF3 should have higher dipole moment than NH3. The dipole moment of NH3 = 1.46D. The dipole moment of NF3 = 0.24D.
How many electron groups are in NF3?
For NF3 , the Lewis Structure will give you something like Nitrogen in the center with 3 bonds to F atoms, and 1 lone pair(I don’t know how to draw structures on here). If you look at the Nitrogen, it has 4 different electron groups around it (3 from the bonds, 1 from lone pair).
What is the hybridization of NF3 and BF3?
THE NF3 IS SP3 HYBRIDISED WHERE AS BF3 IS SP2 HYBRIDISED .
What is the hybridization of NO2?
In the classical sense, NO2 is sp2 hybridized. In general, the Single-electron orbitals are unhybridized, and pure p-orbitals, like in methyl free radicals. However, a Single-electron orbital is known to be hybridized, when the central atom is bonded to the highly electronegative groups or atoms.
How do you determine hybridization?
Here’s what you do: Look at the atom. Count the number of atoms connected to it (atoms – not bonds!) Count the number of lone pairs attached to it.
Add these two numbers together.
If it’s 4, your atom is sp3.If it’s 3, your atom is sp2.If it’s 2, your atom is sp.
Which of the following is polar NF3?
NF3 is trigonal pyramidal in shape. This is because N requires 4 electron pairs to fulfil its octet. NF3 (nitrogen trifluoride) is the polar molecule in this case.
Why is NF3 dipole dipole?
In NF3, F is more electronegative than N. So, all F atoms pull the electrons towards themselves. And, this resultant moment is opposite to the directions of that of the lone pair of electrons on the ‘N’ atom. And, since Dipole Moment is a vector quantity, the net dipole decreases.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
