Is NH3 trigonal planar?
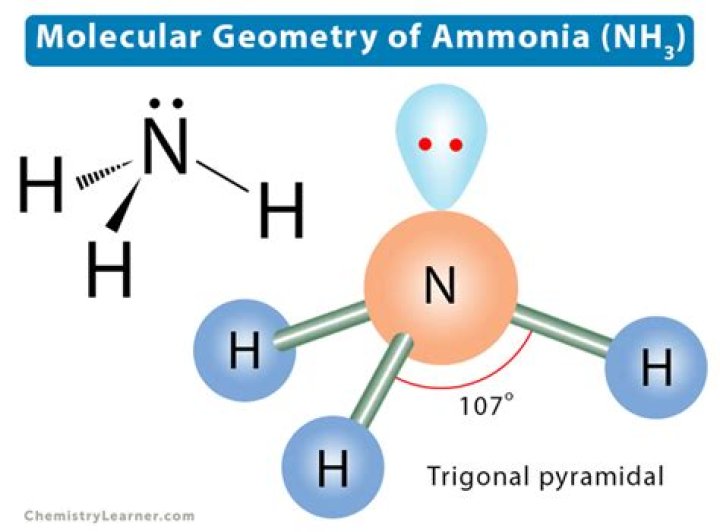
NH3 molecular geometry is trigonal pyramidal. The reason for this is that the nitrogen atom has a single non-bonding lone pair of electrons that serves as a repulsive force on the bonding orbitals. The three hydrogen atoms are bonded to a nitrogen atom in the middle.
Is NH3 trigonal planar?
It is trigonal pyramidal because of the lone pair of electrons associated with the central nitrogen atom.
Why is NH3 tetrahedral?
The NH3 molecule has a tetrahedral geometry shape because it contains three hydrogen atoms. There are three N-H bonds at the NH3 molecular geometry. After linking the three hydrogens and one lone pair of electrons in the tetrahedral form, it maintains the tetrahedral-like structure.
Is NH3 tetrahedral geometry?
Ammonia has 4 regions of electron density around the central nitrogen atom (3 bonds and one lone pair). These are arranged in a tetrahedral shape.
Why is NH3 a trigonal pyramidal?
It has no side effects of its own but can precipitate withdrawal symptoms in addicts. The valence shell electron pair repulsion model assumes that electron pairs repel one another. If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3).
Is NH3 a polar molecule?
Ammonia is polar, the N is the negative end, and the middle of the H’s is the positive end.
Why NH3 has distorted shape?
Among them, three take point in covalent bond formation with three hydrogen atoms and the other two remain unshared as lone pairs. Lone pairs can’t decide the shape of molecules. So, according to VSEPR theory, the shape of the ammonia molecule is distorted tetrahedral as there is no same type of four bonds.
Why does ammonia have a tetrahedral electron geometry but a trigonal pyramidal molecular geometry?
Three of these electron pairs are used as bond pairs which leaves one lone pair of electrons. The lone pair repel more strongly than bond pairs giving tetrahedral arrangement. Since the lone pairs are invincible the shape of ammonia is trigonal pyramidal.
Which is molecular geometry of bf3 molecule is?
The geometry of the BF 3 molecule is called trigonal planar (see Figure 5).
What bond is NH3?
NH3 has a covalent single bond among its nitrogen and hydrogen atoms.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
