Is NH4 a dipole?
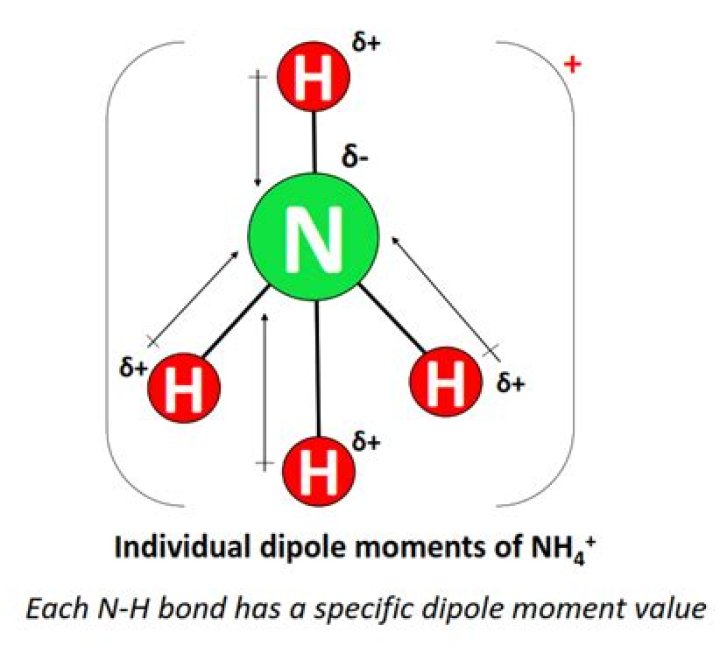
If we talk about the chemical composition of ammonium ion, it consists of 4 hydrogen atoms and a nitrogen atom. … The dipole value of NH4+ ions is also non zero. But due to the symmetrical arrangement of N-H bonds, the dipoles get canceled out by each other resulting in a nonpolar molecule.
Is NH4+ A dipole-dipole interaction?
There are no dipole-dipole forces, nor is hydrogen bonding present (in a vast sea of NH4+ ions you have tons of h-bond donors, but no h-bond acceptors). The only intermolecular forces between NH4+ ions are van der waals (london dispersion forces).
Is NH4+ polar or nonpolar?
Is NH4+ (Specifically the Ammonium Ion) Polar? It is non-polar. The reason for this is because it is tetrahedral, and all Hydrogens are the same. There is no reason for any one hydrogen or proton to be treated differently, so they are all treated the same.
When the ammonium ion, NH4+, is formed, the fourth hydrogen is attached by a dative covalent bond, because only the hydrogen’s nucleus is transferred from the chlorine to the nitrogen. The hydrogen’s electron is left behind on the chlorine to form a negative chloride ion.
Does ammonia have a dipole moment?
NH3 is a polar molecule because, in the NH3 molecule, it has three dipoles because of three bonds and these dipoles do not cancel out each other. They form a net dipole moment.
NO3 is a non-polar molecule because of the symmetrical structure irrespective of the presence of a double bond, which cancels out all the dipole moments inside the molecule. As there is no net dipole moment, there will be no separation of charges between the two ends of the molecule and hence no polarity.
What intermolecular forces are in NO3?
Ion-induced dipole forces – Intermolecular force exist between an ion and a non-polar molecule. Here the charge of the ion creates temporary dipole on non-polar molecule. e.g. I2 & NO3 −.
Is NH4 an acid or base?
The ammonium ion(NH4+) is an acid in nature as it releases the proton when dissolved in an aqueous solution. However, it is a very weak acid having a pH value of 5.5 i.e. near to neutral in the acid-base scale. The proton donating ability of NH4+ makes it mildly acidic.
The bond angles in ammonium ion, NH4^+ are the theoretical tetrahedral bond angles of approximately 109.5 degrees. The ammonium ion is completely symmetrical.
What is the shape of NH4+?
That leaves a total of 8 electrons in the outer level of the nitrogen. There are therefore 4 pairs, all of which are bonding because of the four hydrogens. The ammonium ion has exactly the same shape as methane, because it has exactly the same electronic arrangement. NH4+ is tetrahedral.
Is NH4 a molecule?
Is Ammonium ion (NH4+ ) a molecular ion? Ammonium ion is not a molecular ion because it is formed by the coordinate covalent bond between ammonia (NH3) and hydrogen ion (H+) . Due to lone pair electron ,ammonia is an electron rich specie and can easily donate a pair of electron to electron deficient specie (H+ ion).
Why is NH4 a coordinate covalent bond?
When the ammonia molecule combines with H+ ion by the donation of lone pair electron from N-atom (in ammonia) to H+ ion results in the formation of ammonium ion ,NH4+. Coordinate Covalent bond is a type of covalent bond that forms when one atom donates a pair of electron(Nucleophile) to another atom (Electrophile).
Dipole Moment: The dipole of a molecule is the measure of its polarity. The polar molecule always has its dipole equals to non zero. And the nonpolar molecules have net dipole equals to zero. Similarly, the dipole of NH4+ (Ammonium ion) is 0 D.
Is ammonia a dipole-dipole?
You know that, ammonia is a polar molecules. it exhibits, dipole-dipole intraction, induced attraction, and London dispersion forces. NH3 is called dipole dipole because nh3 make N-H bond, it directly make hydrogen bonding.
Is ammonia a dipole?
The ammonia molecule has a trigonal pyramidal shape as predicted by the valence shell electron pair repulsion theory (VSEPR theory) with an experimentally determined bond angle of 106.7°. This shape gives the molecule a dipole moment and makes it polar.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
