Is nitrogen dioxide a covalent compound?
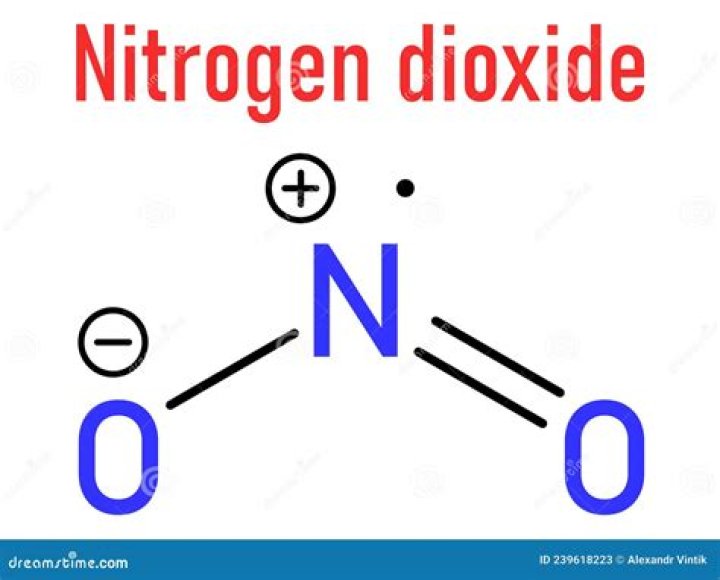
The nitrogen dioxide is a covalent compound where one nitrogen is the central atom which is bonded to two oxygen atoms, where one oxygen atom is bonded by a single bond and other oxygen atom by a double bond.
Is nitrogen oxide a covalent compound?
Nitrogen dioxide is a covalent compound.
What type of compound is nitrogen dioxide?
Nitrogen Dioxide (NO2) is one of a group of highly reactive gases known as oxides of nitrogen or nitrogen oxides (NOx).
Nitrogen Dioxide (NO2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. At room temperatures, nitrogen dioxide is a reddish-brown gas that has a density of 1.8 g/dm3.
Is nitrogen dioxide ionic or covalent compound?
The nitrogen dioxide is a covalent compound where one nitrogen is the central atom which is bonded to two oxygen atoms, where one oxygen atom is bonded by a single bond and other oxygen atom by a double bond.
Nitric oxide (NO) and nitrogen dioxide (NO2) are two gases whose molecules are made of nitrogen and oxygen atoms. Nitrogen dioxide is a strong oxidizing agent, and is thus very reactive with other compounds.
Is nitrogen a compound?
Nitrogen is an element that can combine with itself or with other elements to make different compounds. For instance nitrogen gas, N2, is a compound made when two nitrogen atoms form a chemical bond. It makes up about 80% of the atmosphere, while oxygen gas, O2, makes up a little less than 20% of the atmosphere.
Is NO2 a compound or molecule?
It is a molecular compound as it is composed of two different elements bonded to one another.
The given is composed of nitrogen and oxygen, bonded chemical, making it a compound. Since nitrogen dioxide is a compound, thus it is a pure substance.
Why is nitrogen oxide covalent and not ionic?
NO2 is a covalent compound because the bond is formed between one nitrogen and two oxygen atom by the sharing of electrons. Also, the difference of electronegativity between nitrogen and oxygen atoms on the Pauling scale is not big enough to make an ionic bond in the NO2 compound.
What is the covalent of nitrogen in NO2?
N2O5 has following structure: From the above structure, it is clear that Nitrogen atom is sharing its electrons with oxygen atom. Nitrogen shares it four pair of electrons with oxygen, therefore nitrogen covalency is four (4).
Is NO2 ionic covalent or metallic?
NO2 or nitrogen dioxide is a covalent compound. NO2 is comprised of nitrogen and oxygen which are both highly electronegative chemical elements.
sol. H2 is a covalent, diatomic, molecule with a sigma covalent bond between two hydrogen atoms. I HOPE IT HELPFUL FOR YOU.
Is co2 an ionic compound?
No, CO2 is not an ionic compound. Meanwhile, CO2 is a compound that is formed between two non-metal atoms (carbon and oxygen) thus giving it a covalent nature. In CO2 one carbon atom will share its four electrons with two electrons from each of the oxygen atoms.
Which of the following compounds are ionic and which are covalent?
The ionic compounds are sodium chloride, ammonium chloride and magnesium chloride. The covalent compounds are urea, cane sugar, hydrogen chloride, carbon tetrachloride, ammonia and alcohol.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
