Is no2 a structure?
The NO2 Lewis structure has a total of 17 valence electrons. It’s not common to have an odd number of valence electrons in a Lewis structure. … In the Lewis structure for NO2 the Nitrogen atom is the least electronegative atom and goes at the center of the structure.
What type of structure is NO2?
NO2 (Nitrogen Dioxide) Lewis Dot Structure. Nitrogen Dioxide (NO2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom.
Is NO2 a linear structure?
NO2 is a bent molecule; however, when you remove an electron from it, making it NO2+, the molecule becomes linear due to the loss of a lone electron. In NO2+, there is no repulsion taking place between the two O atoms and the lone electron on the central atom.
You must first draw the Lewis structure for NO2 . You find that you can draw two Lewis structures.
Is NO2 trigonal planar?
In this example, NO2, the Lewis diagram shows nitrogen at the center with one lone electron. Hence the molecule has three electron pairs and is trigonal planar for electron pair geometry.
Why are NO2 and CO2 different shapes? two N=O double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2.
Is NO2 a molecule or compound?
It is a molecular compound as it is composed of two different elements bonded to one another.
Is NO2 an anion?
It is a nitrogen oxoanion, a member of reactive nitrogen species and a monovalent inorganic anion. It is a conjugate base of a nitrous acid.
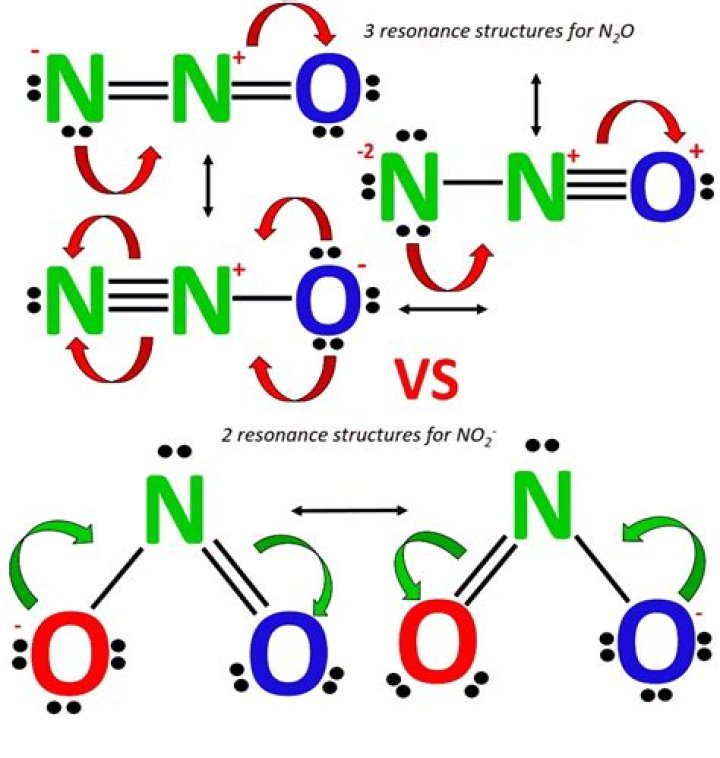
After determining how many valence electrons there are in NO2-, place them around the central atom to complete the octets. There are a total of 18 valence electrons for the Lewis structure for NO2-. Nitrogen is the least electronegative atom in the NO2- lewis structure and therefore goes in the center of the structure.
What is Lewis structure of nitrogen?
Each N is surrounded by two dots and three sticks or lines, representing another 6 electrons in the N2 triple bond. So each N is surrounded by 8 total valence electrons, giving it an octet and making it stable. The two letter N’s in the N2 Lewis structure represent the nuclei (centers) of the nitrogen atoms.
What is the difference between no2 and no2?
The key difference between nitrite and nitrogen dioxide is that the nitrite is an anion whereas the nitrogen dioxide is a molecule. Both nitrite and nitrogen dioxide has the same number of nitrogen and oxygen atoms; one nitrogen atom and two oxygen atoms.
Is NO2 a polar molecule?
Answer: NO2 is polar molecule due to the presence of a lone pair of electrons whose electron-electron repulsion results in a bent structure.
Nitrous oxide is linear.
Which molecule has a linear structure?
Linear molecule is a molecule in which atoms are deployed in a straight line (under 180° angle). Molecules with an linear electron pair geometries have sphybridization at the central atom. An example of linear electron pair and molecular geometry are carbon dioxide (O=C=O) and beryllium hydrideBeH2.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
