Is NO2 Plus or minus?
NO2 is a bent molecule; however, when you remove an electron from it, making it NO2+, the molecule becomes linear due to the loss of a lone electron. In NO2+, there is no repulsion taking place between the two O atoms and the lone electron on the central atom.
Is NO2 positively charged?
NO2 has two bonds on the N and Nitrogen makes three bonds to have a full outer shell. It has one unpaired electron, therefore a positive charge. That unpaired electron can make one more bond to get rid of the positive charge (accept an electron) which was what electrophiles do. Electrophiles all have positive charges.
What is hybridisation of NO2 plus?
So, the species is tetrahedral with sp3 hybridisation. So, the species is linear with sp hybridisation.
It is isoelectronic with CO2, having two N=O. double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2.
What is the bond angle of NO2+?
$NO_2^ + $ has two bond pairs and zero lone pairs so hybridization of $NO_2^ + $is $sp$, and it will have linear geometry. It is clear that for linear geometry bond angle is ${180^ circ }$. Therefore the bond angle of $NO_2^ + $ is${180^ circ }$.
However, the shape of the molecule is described in terms of the positions of the atoms, not the lone electron pairs. Thus, NO2– is said to have a bent shape, not a trigonal planar one. It is assumed that the lone pair is “invisible” when describing the shape of the molecular geometry.
Why is NO2 neutral?
To give a acidic solution the compound should have an ability to release hydronium ion. For that, water should be hydrolyzed and should form hydronium ion. But polarization of atoms of nitrogen dioxide and carbon monoxide is not enough to hydrolyze water. So they are neutral oxides.
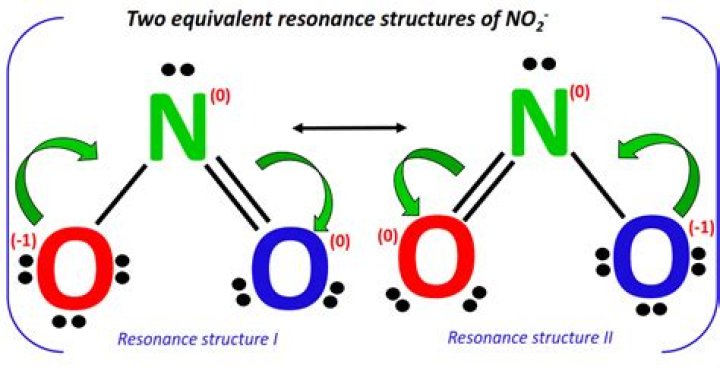
Where is the negative charge in NO2?
The oxygen atom bonded by a single bond has only one electron involved in bonding, and so contains one unpaired electron. Hence, in order to complete its octet it acquires one additional electron. As a result of this, the nitrite ion has an overall charge of -1.
A sodium atom has one electron in its outer shell. A sodium atom can lose its outer electron. It will still have 11 positive protons but only 10 negative electrons. So, the overall charge is +1.
What is the bond order in NO2 plus?
Its bond order is 2 .
What is the geometry of I3 plus?
The molecular geometry of I3- is linear. While there are 3 Iodine atoms, one of the atoms has a negative charge, which further gives 2 bond pairs and 3 lone pairs of electrons.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
