Is p2o5 covalent or ionic?
P2 O5 is covalent. A general clue that a compound is covalent is two nonmetallic elements bonded together. This stems from the electronegativity…
Is P2O5 ionic or covalent bond?
P2O5 (phosphorus pentoxide) Covalent. When two nonmetals or two negatively charged atoms try to form bonds, they always form covalent bonds. Hence, Dinitrogen Pentoxide is not ionic – it is covalent.
Is cro3 ionic or covalent?
Cation in high oxidation state are said to exhibit high polarisibility. Hence, CrO3 is covalent while CrO is ionic.
Potassium iodide (KI) forms an ionic bond. Potassium and iodine have very different electronegativities. The two atoms would form an ionic bond since ionic bonds form between atoms with a large difference in electronegativity (difference>1.7 using the Pauling scale will result in an ionic bond).
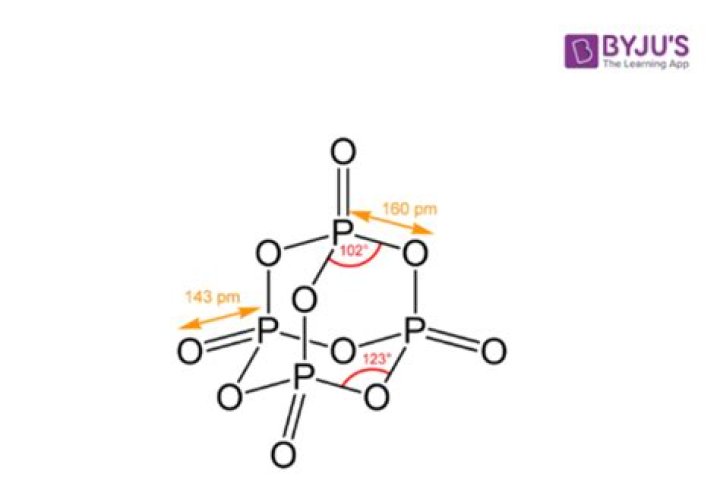
What type of compound is P2O5?
Phosphorus Pentoxide is a chemical compound whose empirical formula is P2O5 and whose molecular formula is P4O10. At ambient temperatures, this compound exists as a white solid that is crystalline in nature. Phosphorus pentoxide is an acid anhydride that is obtained from phosphoric acid.
The nitrogen dioxide is a covalent compound where one nitrogen is the central atom which is bonded to two oxygen atoms, where one oxygen atom is bonded by a single bond and other oxygen atom by a double bond. Total valence electrons present in nitrogen dioxide molecules is 17 (two oxygen atoms present).
Is chromium oxide ionic?
Explanation: The first thing to notice here is that you’re dealing with an ionic compound that contains chromium, Cr , a transition metal, and oxygen, O , a non-metal. The interesting thing about this compound is that it contains a transition metal as its cation, i.e. its positively charged ion.
What is covalent and ionic?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions.
In general, covalent bonds form between nonmetals, ionic bonds form between metals and nonmetals, and metallic bonds form between metals.
Is C12H22O11 ionic or covalent?
Sucrose (table sugar), C12H22O11 is MOLECULAR or COVALENT compound, while sodium chloride (table salt) is _ an IONIC compound. 6. Carbon monoxide, CO, is an example of a diatomic molecule, while ammonia and glucose, NH3 and C6H12O6, are examples of POLYATOMIC molecules.
What type of compound is NH4NO3?
Ammonium nitrate is a chemical compound with the chemical formula NH4NO3. It is a white crystalline solid consisting of ions of ammonium and nitrate. It is highly soluble in water and hygroscopic as a solid, although it does not form hydrates.
Is NH4NO3 covalent bond?
Answer: Its has ionic bond as it exists NH₄⁺ion and NO₃⁻ion. there is covalent bond among nitrogen and oxygen exists.
The cluster cores are surrounded by varying numbers of ammonium nitrate monomer units. Most interesting is the extensive series of negative cluster ions of the form [(NH4NO3)nNO3]-, n g 3. The corresponding positive clusters, [(NH4NO3)nNH4]+, are also very extensive but also include the smallest ions, n ) 1 and 2.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
