Is pbr3 trigonal planar?
According to the VSEPR theory, the PBr3 molecule ion possesses trigonal pyramidal molecular geometry. Because the center atom, phosphorus, has three P-Br single bonds with the three bromine atoms surrounding it. The Br-P-Br bond angle is 101.5 degrees in the trigonal pyramidal PBr3 molecular geometry.
Is pbr3 trigonal planar?
The answer is A. trigonal pyramidal.
Is pbr3 a bond angle?
109.5 degrees | Study.com.
What is the bond angle of pi3?
The bond angles of PF3, PCl3, PBr3 and PI3 are 97o, 100o, 101.5o and 102o respectively. This data shows a gradual increase in the bond angle.
What is the molecular geometry of PBr3 quizlet?
Use VSEPR theory to predict the electron-pair geometry and the molecular geometry of phosphorus tribromide, PBr3. The electron-pair geometry is tetrahedral, the molecular geometry is trigonal-pyramidal.
Why Every molecule has a definite geometry?
(i) All bonded atoms occupy such positions (around the central atom) in space where the repulsive forces between them are minimum. (ii) The molecule attains minimum enthalpy and maximum stability.So molecule has a definite shape or geometry.
What is the molecular geometry of a molecule with 2 outer atoms and 1 lone pair on the central atom?
Nathan L. This is a bent molecule.
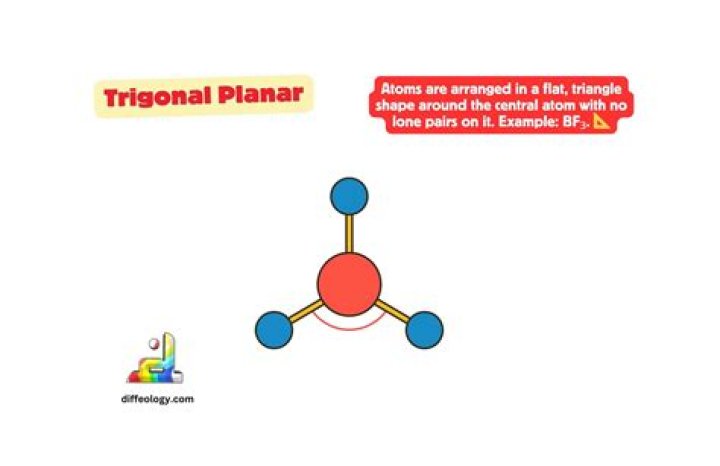
What is trigonal planar in chemistry?
In chemistry, trigonal planar is a molecular geometry model with one atom at the center and three atoms at the corners of an equilateral triangle, called peripheral atoms, all in one plane. In an ideal trigonal planar species, all three ligands are identical and all bond angles are 120°.
What is the molecular geometry of a molecule that has three bonding and two non bonding domains?
The molecular geometry of a molecule that has three bonding and two non-bonding domains is T-shaped.
What is the molecular geometry of PBr5?
PBr5 ‘s molecular geometry is trigonal bipyramidal.
Is PI3 tetrahedral?
In order to determine the molecular shape of the PI3 molecule using VSEPR Theory, its Lewis structure must first be determined. P has a steric number of 4 and a coordination number of 3, which means that it has a tetrahedral electron geomerty and a trigonal planar molecular geometry.
How many bonds does PI3 have?
Due to P having 3 bonding pairs and 1 lone pair, the hybridization is sp3 s p 3 to accommodate the 4 pairs of electrons.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
