Is SF4 seesaw or trigonal bipyramidal?
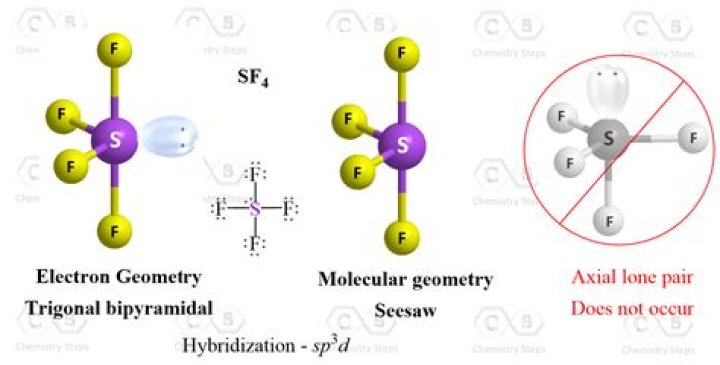
These are arranged in a trigonal bipyramidal shape with 102° F-S-F bond angles between the equatorial fluorine atoms and 173° between the axial fluorine atoms. The lone pair takes an equatorial position because it demands more space than the bonds.
Why SF4 has a seesaw shape?
This shape is caused by a lone pair of electrons on the central atom. An example of a seesaw shaped molecule is sulfur tetrafluoride, or SF4. The equatorial plane fluorine atoms form an angle of 89 degrees, and the angle formed from the equatorial plane fluorine atoms to the axial plane fluorine atoms is 103 degrees.
Is SF4 trigonal planar?
Sulfur tetrafluoride has 5 regions of electron density around the central sulfur atom (4 bonds and one lone pair). These are arranged in a trigonal bipyramidal shape with 102° F-S-F bond angles between the equatorial fluorine atoms and 173° between the axial fluorine atoms.
The molecular geometry of SF4 according to its molecular formula and hybridization is trigonal bipyramidal. The shape generally resembles a see-saw and this shape is due to the repulsion in bonding and lone pairs of electrons.
What is the geometry and shape of SF4?
SF4 molecular geometry is see-saw with one pair of valence electrons. The nature of the molecule is polar. These atoms form a trigonal bipyramidal shape.
The electrons follow this pattern of arrangement following the VSEPR rule to minimize the repulsion forces between the lone pairs of electrons to maximize the molecule’s stability. Hence, SF4 has a trigonal bipyramidal molecular geometry.
Is SF4 a seesaw?
An example of a seesaw shaped molecule is sulfur tetrafluoride, or SF4. Sulfur is the central atom, two fluorine atoms are on the equatorial plane, and two are on the axial plane.
Which of the following species is seesaw in shape?
SF2Cl2 has a see-saw shape.
In the compound SF2Cl2, sulphur is sp3d hybridized and has four coordination number due to its four bonds, two with chlorine and two with fluorine. SF2CL2 is said to be in see saw shape because it looks like a see saw.
It has a square planar geometry.
Is SF4 trigonal bipyramidal?
The SF4 Lewis structure is the combination of 34 valence electron and 5 electron pairs around the Sulfur, where there are 4 bonding pairs and 1 lone pair. This electron arrangement is called ‘Trigonal Bipyramidal’.
What bond is SF4?
The type of bond for SF4 is a covalent bond. The Sulfur tetrafluoride consists of 1 central atom, Sulfur, and 4 Fluorine atoms with 34 valence electrons. It forms four covalent bonds and one lone pair of electrons, as seen when using Lewis Structure.
Are SF4 SF bonds polar?
SF4 (Sulfur tetrafluoride) is polar in nature as sulfur atom consists of a lone pair on it due to which the shape of the molecule becomes asymmetric ie; seesaw.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
