Is tetrahedral CCl4 polar or nonpolar?
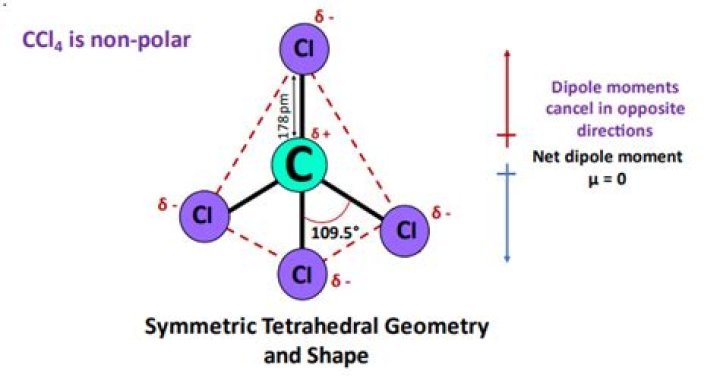
The molecule of CCl4 is nonpolar in nature because of its symmetrical tetrahedral structure. However the C-Cl bond is a polar covalent bond, but the four bonds cancel the polarity of each other and form a nonpolar CCl4 molecule.
Is tetrahedral polar or nonpolar?
Any 100% symmetrical tetrahedral molecule will be nonpolar.
Tetrahedral molecules have no nonbonding electron pairs and all identical bond angles. Therefore, the only way they can be asymmetric is if one atom is different from the rest.
Is the bond between CCl4 polar or nonpolar?
Explanation: We could represent the polarity of an individual C−Cl bond as δ+C−Clδ− in that chlorine is more electronegative than carbon, and charge separation, i.e. polarity occurs. And thus the C−Cl bond is POLAR.
In geometry, a tetrahedron (plural: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular faces, six straight edges, and four vertex corners.
Why is CCl4 a nonpolar molecule but PCl3 is a polar molecule?
Why is CCl4 a nonpolar molecule, but PCl3 is a polar molecule? Because the molecule CCl4 has a tetrahedral shape, the four C−Cl dipoles cancel, which makes CCl4 a nonpolar molecule.
CCl4 has a tetrahedral geometry with bond angles of 109.5 °.
What is the geometry of CCl4?
CCl4 Molecular Geometry
The central atom of this compound is Carbon. Carbon is bonded to 4 Chlorine atoms. The Carbon atom takes a central place and the rest Chlorine atoms are placed around it. Four bonded pairs make the geometry of CCl4 to be Tetrahedral.
Is CCl4 bent or linear?
In this compound carbon is present in the central position and rest all the chlorine atoms are placed around it. As the central metal atom has four bonded pairs and shows $s{{p}^{3}}$hybridization therefore the shape of the molecule is tetrahedral.
More specifically speaking, carbon tetrachloride is a nonpolar covalent compound because the electrons shared by the carbon and chlorine atoms are nearly at the center of the bond. Therefore, the carbon tetrachloride ($CC{{l}_{4}}$) is a covalent compound.
Why is CCl4 a nonpolar molecule?
CCl4 that is carbon tetrachloride is nonpolar because all the four bonds are symmetrical, and they are they extended in all the directions. This makes it easy for the dipole moments in each direction to cancel out.
Why does CCl4 have polar bonds?
CCl4 molecule has one carbon atoms attached to the four chlorine atom. It can be understood because Carbon and Chlorine atom have a different value of electronegativity ie (C= 2.55, Cl= 3.16). Making the C-CL bond a polar covalent bond. Therefore all four C-Cl bonds have a dipole moment value as well.
What is the electronegativity of CCl4?
The four bonds of carbon tetrachloride (CCl4) are polar, but the molecule is nonpolar because the bond polarity is canceled by the symmetric tetrahedral shape. The electronegativity for C is 2.5 and Cl is 3.0, resulting in a polar covalent bond.
In a tetrahedral molecule, there is one central atom bonded to four surrounding atoms with no lone electron pairs. The bonds form angles of 109.5°. Some examples of tetrahedral molecules include the ammonium ion, methane ion, and phosphate ion.
Which of the following is nonpolar?
Any of the homonuclear diatomic elements: H2, N2, O2, Cl2 (These are truly nonpolar molecules.) Carbon dioxide – CO. Benzene – C6H. Carbon tetrachloride – CCl.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
