Is water covalent or ionic?
The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).
Is water a covalent?
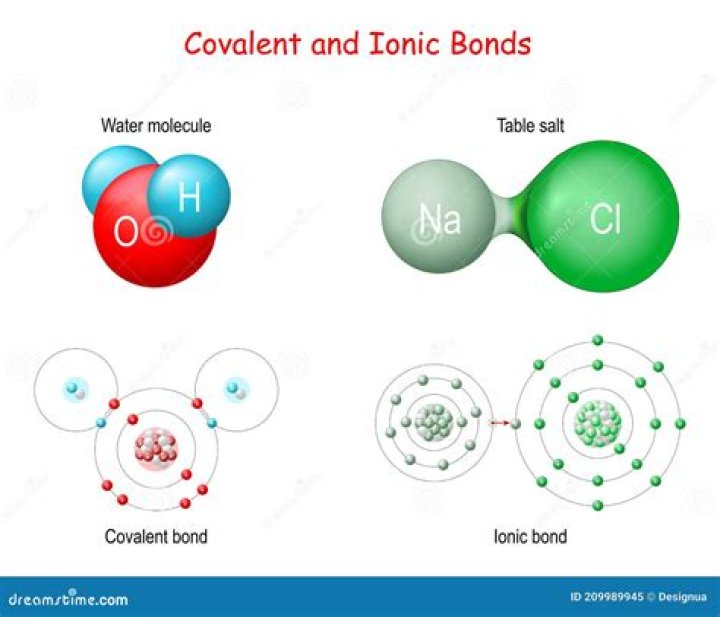
A water molecule consists of two atoms of hydrogen linked by covalent bonds to the same atom of oxygen. Atoms of oxygen are electronegative and attract the shared electrons in their covalent bonds.
Why is water not ionic?
Water is said to belong to the covalent category. In other words, water is made up of covalent bonds and not ionic bonds. This primarily because the water molecule has an unsymmetrical shape. The positive charge is on the hydrogen pole (side) and the negative charge is on the oxygen pole (side).
Water is a polar molecule. While the overall charge of the molecule is neutral, the orientation of the two positively charged hydrogens (+1 each) at one end and the negatively charged oxygen (-2) at the other end give it two poles.
What bond is between water molecules?
In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. The attraction between individual water molecules creates a bond known as a hydrogen bond.
Strong linkages—called covalent bonds—hold together the hydrogen (white) and oxygen (red) atoms of individual H2O molecules. Covalent bonds occur when two atoms—in this case oxygen and hydrogen—share electrons with each other.
Why is H2O covalent?
In the case of water, the electronegativity of oxygen is 3.5, while that of hydrogen is 2.1. In summary, water has a covalent bond because of the nature of oxygen and hydrogen — they share electrons to attain stability, and their electronegativities are close enough for their bond to be considered covalent.
Is water an element?
Answer. Water is a compound. It contains more than one element: hydrogen and oxygen atoms are joined together; as illustrated in the video clip Elements and Compounds, above.
An ionic compound consists of two oppositely charged ions. Water, on the other hand is a polar solvent; the electronegativity difference between oxygen and hydrogen is high which is why water has a positive pole of H and a negative pole O (water is H2O ).
Why is water covalent bond?
A water molecule consists of one oxygen atom and two hydrogen atoms held together by a covalent bond. In this case the oxygen atom shares one electron with each hydrogen atom. This means that the oxygen atoms has a small net positive charge and the hydrogen atom has a small net negative charge.
Is water a solvent?
Water is called the “universal solvent” because it is capable of dissolving more substances than any other liquid. It is water’s chemical composition and physical attributes that make it such an excellent solvent.
Why is water considered polar?
The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms. When a neutral molecule has a positive area at one end and a negative area at the other, it is a polar molecule.
The scientific name for water is H2O. It is called H2O because it has two atoms of hydrogen (H) and one atom of oxygen (O). There are millions of these molecules in one drop of water. The form water takes depends on the movement of the water molecules.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
