name of group 13 on the periodic table, check these out | Why is group 13 called?
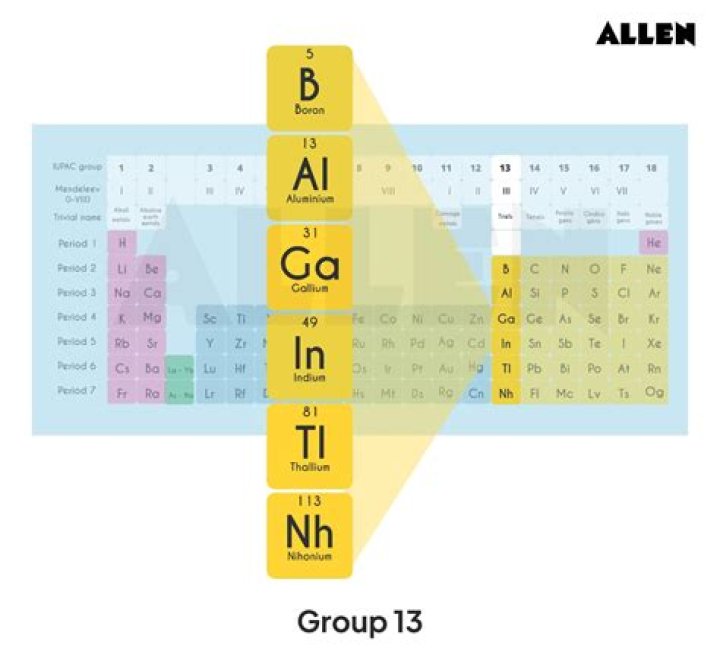
boron group element, any of the six chemical elements constituting Group 13 (IIIa) of the periodic table. The elements are boron (B), aluminum (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh).
Why is group 13 called?
The boron group is sometimes called group 13 of the periodic table since the first member at the top of the group is boron (B). Even in this community, Boron is the only metalloid. All the metals are the other four elements in the group: aluminium (Al), gallium (Ga), indium (In), and thallium (Tl).
Where is group 13 on the periodic table?
Group 13 is sometimes referred to as the boron group, named for the first element in the family. These elements are–not surprisingly–located in column 13 of the periodic table. This group includes boron, aluminum, gallium, indium, thallium, and ununtrium (B, Al, Ga, In, Tl, and Uut, respectively).
What is group 13 and 14 on the periodic table called?
Group 13 is called the boron group, and boron is the only metalloid in this group. The other group 13 elements are metals. Group 14 is called the carbon group. This group contains two metalloids: silicon and germanium.
What is the 13 on the periodic table?
Aluminium – Element information, properties and uses | Periodic Table.
What do Group 13 elements have In common?
All group 13 elements have ns 2np 1 valence electron configurations, and all tend to lose their three valence electrons to form compounds in the +3 oxidation state. The heavier elements in the group can also form compounds in the +1 oxidation state formed by the formal loss of the single np valence electron.
How do the Group 13 elements differ from the Group 14 elements?
All group 13 elements have three valence electrons and are fairly reactive. All are solids at room temperature. Group 14 is called the carbon group. Carbon (C) is a nonmetal.
Are group 13 elements metals?
The boron family contains elements in group 13 of the periodic talbe and include the semi-metal boron (B) and the metals aluminum (Al), gallium (Ga), indium (In), and thallium (Tl).
Which of the following is NOT group 13?
All the elements of group 13 are also called the boron family. The elements of the 13th group element are boron, aluminium, gallium, indium, and thallium. They all are metallic in nature except boron which is a metalloid.
What is the Valency of group 13 elements?
The valency of group 13 elements is 1 and the valency of oxygen is 2 and to satisfy the combining capacity of group 13 elements, 3 of halogens are required. Thus, the molecular formula will be of form MX3. For example: AlCl3,BF3 etc.
What are groups 13/18 called on the periodic table?
Group 18 elements are called noble gases. Groups 1, 2, and 13-18 are the representative elements (or main-group elements). Groups 3-12 are called the transition metals.
What are groups 13/16 called on the periodic table?
Groups 13–16 fall between the transition metals (in groups 3–12) and the nonmetals called halogens (in group 17). What Are Metalloids? Metalloids are the smallest class of elements, containing just six members: boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), and tellurium (Te).
Which of the Group 13 elements is the largest?
Thallium is the heaviest element. T.Boron has the highest melting point. T.Electron potential increases going down the group. T.Thallium has the lowest ionization energy. F.All of the above are correct. T.
Who discovered element 13?
The name for element 13 has been either aluminum or aluminium. We can blame the English chemist, Sir Humphy Davy, for the confusion. Davy initially called the element alumium in 1807, from the mineral alumina. Davy changed the name to aluminum and then finally to aluminium in 1812.
Why is aluminum’s atomic number 13?
Explanation: Aluminum has (by definition) 13 protons, 13 positively charged particles. Because aluminum is neutral (as is all matter), it therefore has 13 negatively charged particles, electrons. Aluminum has an atomic mass of about 27 amu; how many neutrons are present in the aluminum nucleus?
What element is in Group 13 and Period 2 of the periodic table?
The element at the position of period 2 and group 13 is boron, which is written as B, which is a metalloid. In period 2, the number of shells filled with electrons of all the elements is 2.
Which group 13 element that never occurs In Free State?
Gallium (Ga; Mendeleev’s eka-aluminum) was discovered in 1875 by the French chemist Paul Émile Lecoq de Boisbaudran during a systematic search for Mendeleev’s “missing” element in group 13. Group 13 elements are never found in nature in their free state.
Which of these group 13 elements has the smallest atomic radius?
Out of the following group 13 elements, element with smallest atomic radius is. Out of Al, Ga, In and Tl element with smallest atomic radius is GA.
Is group 13 transition metals?
Typically the elements of the post-transition metals include any metal in groups 13, 14, and 15 which are aluminum, gallium, indium, tin, thallium, lead, and bismuth. Polonium is often classified as a post-transition metal as well.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
