news
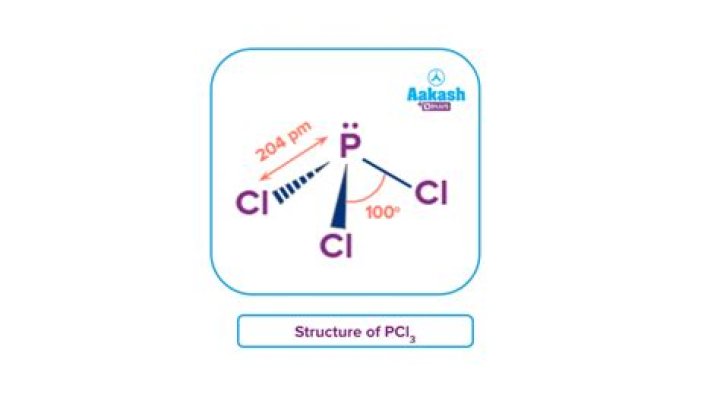
O2 – oxygen.Cl2 – chlorine.PCl3 – phosphorus trichloride.CH3CH2OH – ethanol.O3 – ozone.H2 – hydrogen.H2O – water.HCl – hydrogen chloride.”
Written by Ella Bryant — 0 Views
What are examples of covalent bonds?
Examples of Covalent Bond:
Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O. Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure. Vulcanized rubber. Another example is vulcanized rubber.
Which of these is an example of a covalent compound?
together by sharing electrons (covalent bonding). Examples are water, which contains H2O molecules; methane, which contains CH4 molecules; and hydrogen fluoride, which contains HF molecules.
What is not a covalent bond?
Which compound is highly covalent?
Applying the second rule we get to know that Iodide ion(I−) size is the largest thereby increasing the covalent character of the compound. The compound with the highest covalent character is CaI2.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
