the reversible isochoric change, check these out | Can isochoric be reversible?
Can isochoric be reversible?
Yes they are reversible because in isothermal heat is transferred at constant temp and in adiabatic there is no any heat transfer.
What is an Isochoric change?
An isochoric process in the context of thermodynamics is a term used to describe a situation where the volume of a system remains constant. For this process to be possible, the ideal gas must be in a rigid container that doesn’t change in volume or let any molecules escape.
Is an isobaric process reversible?
Most reactions are irreversible. But, yes isobaric processes are mostly reversible because work done is proportional to volume and not pressure as in isobaric process pressure is constant.
What is the work done in isochoric process?
Whether the pressure change is positive or negative, the work done by the gas in an isochoric process is zero. Work done by a gas in a process in which PVn = constant, where n is also constant.
What is reversible adiabatic?
Reversible adiabatic process is also called an Isentropic Process. It is an idealized thermodynamic process that is adiabatic and in which the work transfers of the system are frictionless; there is no transfer of heat or of matter and the process is reversible.
What is the difference between reversible and irreversible process?
A reversible process is one in which both the system and its environment can return to exactly the states they were in by following the reverse path. An irreversible process is one in which the system and its environment cannot return together to exactly the states that they were in.
What is isobaric change?
In thermodynamics, an isobaric process is a type of thermodynamic process in which the pressure of the system stays constant: ΔP = 0. The heat transferred to the system does work, but also changes the internal energy (U) of the system.
What is isochoric and isobaric process?
1) Isochoric process is a chemical process that takes place in a thermodynamic system under a constant volume. 1) An isobaric process is a chemical process that takes place in a thermodynamic system under constant pressure. 2) There is no change in volume ΔV=0. 2) The change in pressure or ΔP=0.
What happens Isochoric expansion?
An isobaric process occurs at constant pressure. An isobaric expansion of a gas requires heat transfer to keep the pressure constant. An isochoric process is one in which the volume is held constant, meaning that the work done by the system will be zero. The only change will be that a gas gains internal energy.
Is reversible process possible?
No, it’s not possible that one process can be seen as reversible as well as irreversible in different reference perspectives. When any process happen then there is change in the state of the system and depending upon the boundary between system and surrounding, sharing of mass, work and heat is happened.
What is reversible and irreversible process thermodynamics?
In terms of thermodynamics, a reversible process is where the participants go back to its initial form by inculcating minor or negligible changes in their surroundings. Contrarily, an irreversible process is a naturally occurring phenomenon, which does not go back to its original state.
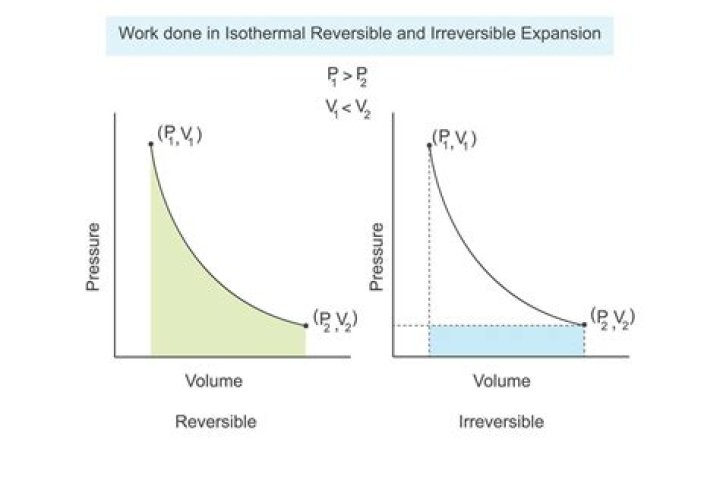
What is isothermal reversible expansion?
Hence, a reversible isothermal expansion is an infinitely-slow increase in volume at constant temperature. For an ideal gas, whose internal energy U is only a function of temperature, we thus have for the first law of thermodynamics: ΔU=qrev+wrev=0.
Why the work done is zero in isochoric process?
As the gas inside the spray can heats up, its pressure increases, but its volume stays the same (unless, of course, the can explodes). Because volume is constant in an isochoric process, no work is done.
What is the equation of isochoric process?
14.2 Isochoric Process
l 12 = ∫ 1 2 p d v ≡ 0. Thus, by the first law of thermodynamics, the change in the internal energy of the system is equal to the heat added to the system, (14.10) (14.11)
What is zero for an isochoric process?
An isochoric process is a process that occurs at constant volume. This means that the change in the volume is zero.
Which of the following is reversible?
Melting, boiling, evaporation, freezing, condensation, and disintegration are examples of reversible transformations. Melting wax, freezing ice, and boiling water that evaporates as steam and condenses back to water are a few examples. Hence option C is correct.
What is the entropy change in a reversible adiabatic process?
The entropy change in an adiabatic process is zero.
Why is adiabatic expansion reversible?
An adiabatic (zero heat exchanged with the surroundings) process is reversible if the process is slow enough that the system remains in equilibrium throughout the process.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
