titrating a weak acid with a strong base, check these out | What happens when you titrate a weak acid with a strong base?
What happens when you titrate a weak acid with a strong base?
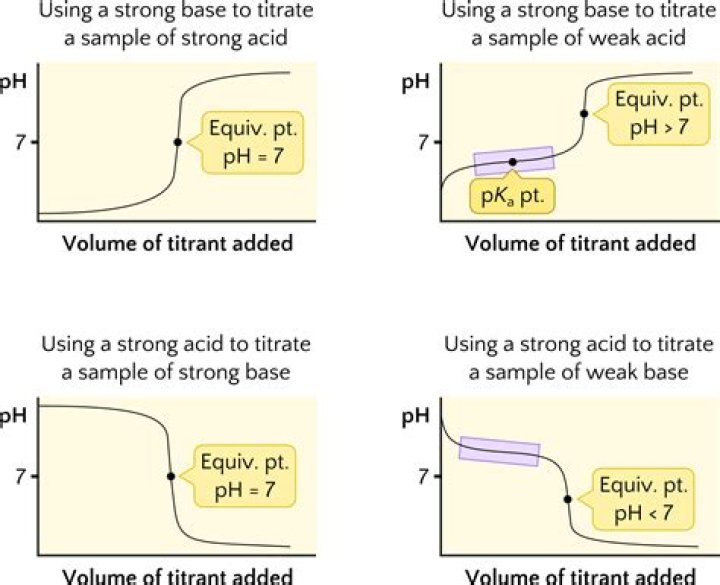
In a weak base-strong acid titration, the acid and base will react to form an acidic solution. A conjugate acid will be produced during the titration, which then reacts with water to form hydronium ions. This results in a solution with a pH lower than 7.
What happens when you titrate a weak acid with a weak base?
For the titration between a weak acid and a weak base, there is no sharp change of pH at the equivalence point. The pH changes gradually over a range of pH values.
Can a buffer be made by titrating a weak acid with a strong base?
A weak acid alone is not a buffer, because there aren’t appreciable quantities of the conjugate base. Buffers cannot be made from a strong acid (or strong base) and its conjugate.
When we titrate a weak acid with a strong base the pH at the equivalence point will be?
POINT OF EMPHASIS : The equivalence point for a weak acid-strong base titration has a pH > 7.00.
What happens if you add a strong acid to a weak acid?
It doesn’t really “react”, a weak acid acts as a weak base when combined with strong acid. The resulting pH will be lower than the weak acid but higher than the strong acid.
When titrating a strong acid with a strong base What is the equivalence point?
In a strong acid-strong base titration, the acid and base will react to form a neutral solution. At the equivalence point of the reaction, hydronium (H+) and hydroxide (OH-) ions will react to form water, leading to a pH of 7.
Why is a strong acid or strong base preferred over weak acid or weak base for acid-base titration?
When doing a titration of a strong acid with a strong base or a strong base with a strong acid it is generally quite easy to detect the endpoint of the titration since the pH will remain very high or very low for most of the titration since the acid or base that is being titrated is virtually 100% ionized and thus has
What is the difference between a strong acid-strong base titration and a weak acid-strong base titration?
In a strong acid-strong base titration, the equivalence point is reached when the moles of acid and base are equal and the pH is 7. In a weak acid-strong base titration, the pH is greater than 7 at the equivalence point. In a strong acid-weak base titration, the pH is less than 7 at the equivalence point.
Can a weak acid and weak base make a buffer?
A buffer is simply a mixture of a weak acid and its conjugate base or a weak base and its conjugate acid. Buffers work by reacting with any added acid or base to control the pH.
What happens when a strong acid or strong base is added to a buffer system?
When a strong acid (H3O+) is added to a buffer solution the conjugate base present in the buffer consumes the hydronium ion converting it into water and the weak acid of the conjugate base. This results in a decrease in the amount of conjugate base present and an increase in the amount of the weak acid.
Why are strong acids and bases Bad buffers?
So to answer your question, a buffer must contain something that is only weakly reactive, and can react further when the need is present. A strong acid/base will totally react, so there is nothing left over to do any buffering.
When titrating a strong acid with a strong base approximately where would the pH be observed when reaching the equivalence point?
The pH at the equivalence point of the titration of a strong acid with a strong base is 7.0.
Which method is used to determine endpoint of the titration between weak acid and weak base?
In an acid-base titration involving strong base and a weak acid, methyl orange can be used as an indicator.
How do you convert strong acids to weak acids?
Buffers convert a strong acid into a weak acid and convert a strong base into a weak base. Buffers work because they consist of a weak acid and its conjugate base (or a weak base and its conjugate acid). Example: A buffer solution contains both acetic acid and its conjugate base (acetate).
What happens when an acid reacts with a base?
The reaction of an acid with a base is called a neutralization reaction. The products of this reaction are a salt and water. In this traditional representation an acid–base neutralization reaction is formulated as a double-replacement reaction.
Does adding strong acid to weak acid decrease pH?
If we add strong acid to weak acid solution, it will increase H3O+ concentration in very large amount. Now equilibrium of weak acid try to keep equilibrium constant unchanged. So concentrations of H3O+ and B- should be decreased. So, dissociation of weak acid should decrease furthermore.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
