What are the allowable spin quantum numbers?
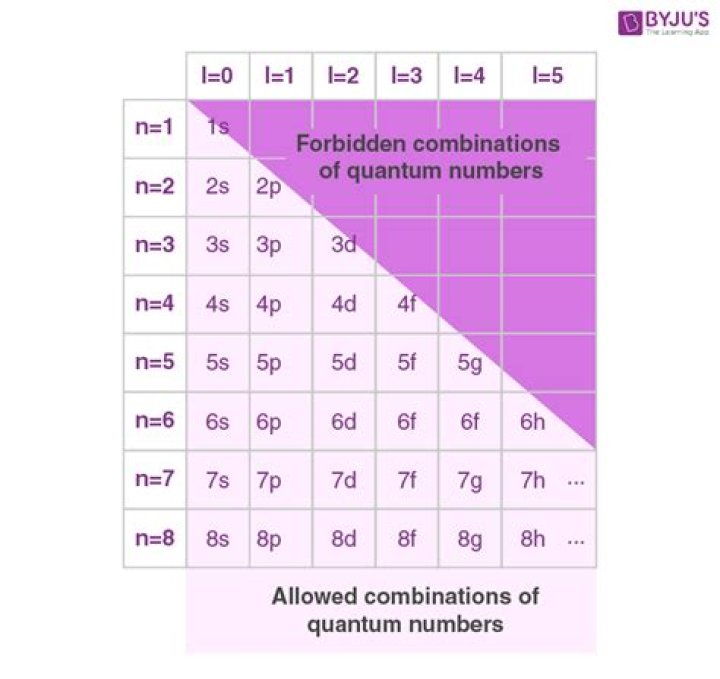
The angular quantum number (l) can be any integer between 0 and n – 1. If n = 3, for example, l can be either 0, 1, or 2. The magnetic quantum number (m) can be any integer between -l and +l. If l = 2, m can be either -2, -1, 0, +1, or +2.
What are the allowable spin quantum numbers?
The three quantum numbers (n, l, and m) that describe an orbital are integers: 0, 1, 2, 3. The principal quantum number (n) cannot be zero. The allowed values of n are therefore 1, 2, 3, 4 The angular quantum number (l) can be any integer between 0 and n – 1.
Which combination of quantum numbers is not allowed?
Of the set of quantum numbers {n, ℓ, m ℓ, m s}, which are possible and which are not allowed? Spin must be either +1/2 or −1/2, so this set of quantum number is not allowed.
Which of the following is a correct set of quantum numbers for an electron in a 3d orbital?
The possible set of quantum numbers for a 3d electron is: b) n = 3, l = 2, ml = 0, ms = –1/2.
Can electrons have the same four quantum numbers?
No two electrons in the same atom can have the same four quantum numbers. Each quantum number describes a different aspect of the electron and its orbital. These numbers are obtained from the solution of the Schrödinger Equation for atoms in spherical coordinates.
Which set of quantum numbers represents permissible values?
Magnetic quantum number (ml): It gives the orbital orientation and the number of permissible values is (2l+1) with values ranging from – l to + l.
What are the allowed values of L when the principal quantum number is N 5?
What are the allowed values of L when the principal quantum number is N 5? So, n=5 means it is the fifth electron shell. ‘l’ is the azimuthal quantum number and it describes electron subshell (s,p,d etc.). Basically, l=0 corresponds to s orbital, l=1 – p orbital, l=2 – d orbital and l=3 – f orbital.
What are the allowed values of the total spin S for iron?
– The quantum numbers for iron are principal quantum number (n) is 4, azimuthal quantum number (l) = 2 for 3d –orbital, magnetic quantum number (m) is -2, -1, 0 , 1, 2 and spin quantum (s) number is either −12or+12 .
What are quantum numbers what permitted values can these have give the significance of each quantum number?
The principal quantum number is an integer that is the number of the electron’s shell. The value is 1 or higher (never 0 or negative). The angular momentum quantum number is an integer that is the value of the electron’s orbital (e.g., s=0, p=1). ℓ is greater than or equal to zero and less than or equal to n-1.
What is the maximum number of electrons that can be identified with the following set of quantum numbers n 4 L 3?
Fourteen electrons can be represented by the values n = 4 and l = 3.
Which set of quantum numbers is not allowed quizlet?
The disallowed set of quantum numbers is n = 2, l = 0, ml = −1, which does not meet all the rules for acceptable values of quantum numbers because for l = 0, ml can only equal 0. n = 2, l = 1, ml = 0; n = 1, l = 0, ml = 0; and n = 3, l = 2, ml = −2 are all allowed sets of quantum numbers.
Which of the following is a correct set of quantum numbers for an electron in a 5f orbital?
The correct set of quantum numbers is choice “a”. Choice “a” is n = 5, l = 3, ml = +1, which is the 5f orbital
Which one of the following is correct set of quantum numbers?
Hence quantum numbers are 4,0,0, +12 or -12.
Which of the following is the correct set of quantum numbers?
Hence the correct set of quantum numbers is: n=3, l=1, m=+1 or 0 or -1, s=+½ or -½.
Why can’t two electrons have 4 quantum numbers?
For each atom, no two electrons can have all four quantum numbers be the same due to the Pauli exclusion principle. Because of this, each orbital has only two electrons, and they have opposite spin.
Why can’t two electrons have quantum numbers?
Pauli’s Exclusion Principle states that no two electrons in the same atom can have identical values for all four of their quantum numbers. In other words, (1) no more than two electrons can occupy the same orbital and (2) two electrons in the same orbital must have opposite spins (Figure 46(i) and (ii)).
Why can’t two electrons have the same quantum number?
Originally Answered: Why can’t two electrons have the same four quantum numbers, but can have three? This is Pauli’s Exclusion Principle. Electrons are fermions (non-integer spin) so therefore they behave according to the exclusion principle, which says they can’t occupy the same quantum state at the same time.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
