What are the limitations of the particle theory?
The particle model does not take into account:
the size and shape of particles.the space between particles.
What are the limitations of the particle theory?
Particle theory can help to explain melting, boiling, freezing and condensing. (HT only) Limitations of the simple model include that there are no forces between the spheres, and that atoms, molecules and ions are not solid spheres.
Why does the particle model have limitations?
The kinetic theory model assumes that particles collide and bounce off each other. But, in reality, particles often do not simply bounce off each other because they have forces of attraction between them. This affects properties such as melting point .
What are the 3 assumptions of the particle model?
The Particle Model
All substances are made of particles.The particles are attracted to each other (some strongly, others weakly).The particles move around (have kinetic energy).As temperature increases, the particles move more (their kinetic energy increases).
What are the advantages of the particle model?
Scientists find the particle model useful for two reasons. First, it provides a reasonable explanation for the behaviour of matter. Second, it presents a very important idea—the particles of matter are always moving. Matter that seems perfectly motionless is not motionless at all.
What are 3 limitations of the particle model?
The particle model does not take into account:
the size and shape of particles.the space between particles.
What are limitations in chemistry?
Limitations are parts of an experiment that keep the scientist from producing fair and reliable data. Even a very well planned out experimental procedure can lead to “mistakes” and produce less than perfect data.
What are the limitations of using a dot and cross diagram?
Dot and Cross Diagrams
Disadvantages: Fails to illustrate the 3D arrangements of the atoms and electron shells. Doesn’t indicate the relative sizes of the atoms.
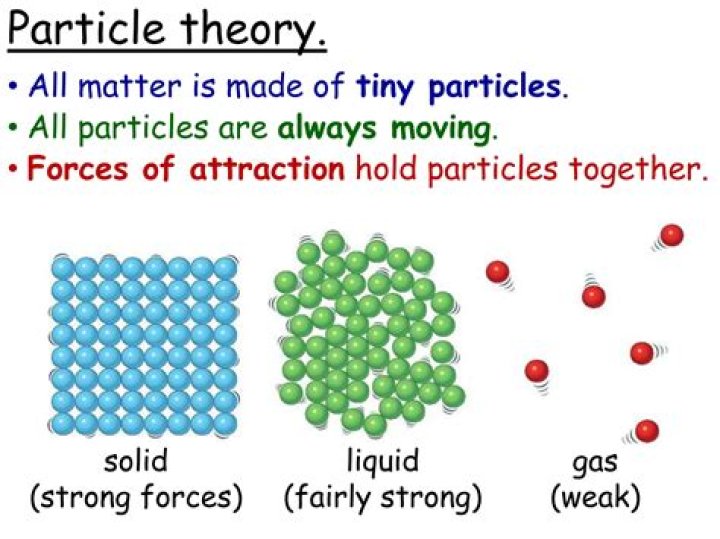
What is the particle model of matter?
The particle theory of matter is a model that describes the arrangement and movement of particles in a substance. The model is used to explain the physical properties of solids, liquids and gases.
What are the strength of particle theory?
Particle theory helps to explain properties and behaviour of materials by providing a model which enables us to visualise what is happening on a very small scale inside those materials. As a model it is useful because it appears to explain many phenomena but as with all models it does have limitations.
What are the 6 rules of particle theory?
3.2 state the postulates of the particle theory of matter (all matter is made up of particles; all particles are in constant motion; all particles of one substance are identical; temperature affects the speed at which particles move; in a gas, there are spaces between the particles; in liquids and solids, the particles
What are the rules of particle theory?
All matter is composed of tiny indivisible particles too small to see. These particles do not share the properties of the material they make up. There is nothing in the space between the particles that make up matter. The particles which make up matter are in constant motion in all physical states.
What are the 4 points of the particle model of matter?
Understand the macroscopic evidence for each of the four basic principles of the particle model of matter:
Matter is made of tiny particles.There is empty space between the particles.The particles are in constant motion.There are forces that act between the particles.
Why is water an exception in the particle model of matter?
Water behaves differently to most other substances because, in its solid state (ice), its particles are less densely packed than in its liquid state. This is why ice floats.
What is the importance of studying the particle arrangement of matter?
It is important to understand the particle nature of matter. The particles that make up matter are not ‘small bits of solid’ or ‘small drops of liquid’ but atoms and molecules. The physical characteristics of those atoms and molecules decide its state.
What are the 5 points of particle theory?
Terms in this set (5)
Particles have space between them. Particles are always moving. Particles move faster and get farther apart when heated. Particles are attracted to each other.
What state is sulfur at 444 C?
Sulphur has a Boiling Point of 444.6°F, meaning at 444.6°F it will turn to a Gas.
Are particles solid spheres?
Particle aren’t spherical. They are 3-dimensional probability waves, with lobes and poles and quantum fuzziness all over. The idea of a point particles started with the Rutherford experiment, last century.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
